Last Updated: March 2026 | v1.0
Semaglutide is a synthetic glucagon-like peptide-1 (GLP-1) receptor agonist that has become one of the most extensively studied compounds in metabolic research. Originally developed for glycemic control research, semaglutide has generated significant scientific interest for its observed effects on energy metabolism, appetite regulation, and body weight reduction in both preclinical and clinical studies. With a molecular weight of approximately 4,113 Da, semaglutide is a modified 31-amino-acid peptide engineered with a C-18 fatty acid chain that extends its half-life by binding to albumin in circulation. This structural modification gives semaglutide a plasma half-life of approximately 7 days — dramatically longer than native GLP-1, which has a half-life of only 2-3 minutes. Published research on semaglutide spans hundreds of peer-reviewed papers, including multiple large-scale randomized controlled trials. This guide consolidates the mechanism of action, key published findings, and standard laboratory reconstitution protocols. All products and information are provided for laboratory and research purposes only.
Quick Facts: Semaglutide
- Classification: GLP-1 receptor agonist (incretin mimetic)
- Molecular weight: ~4,113 Da
- Half-life: ~7 days (vs 2-3 minutes for native GLP-1)
- Key modification: C-18 fatty acid chain enables albumin binding and extended circulation
- Primary research areas: Glycemic control, energy metabolism, appetite regulation, body weight, cardiovascular outcomes
Use the free reconstitution calculator for exact syringe unit calculations, or browse the semaglutide reference page for dosing tables and protocol data.
What Is the Mechanism of Action of Semaglutide?
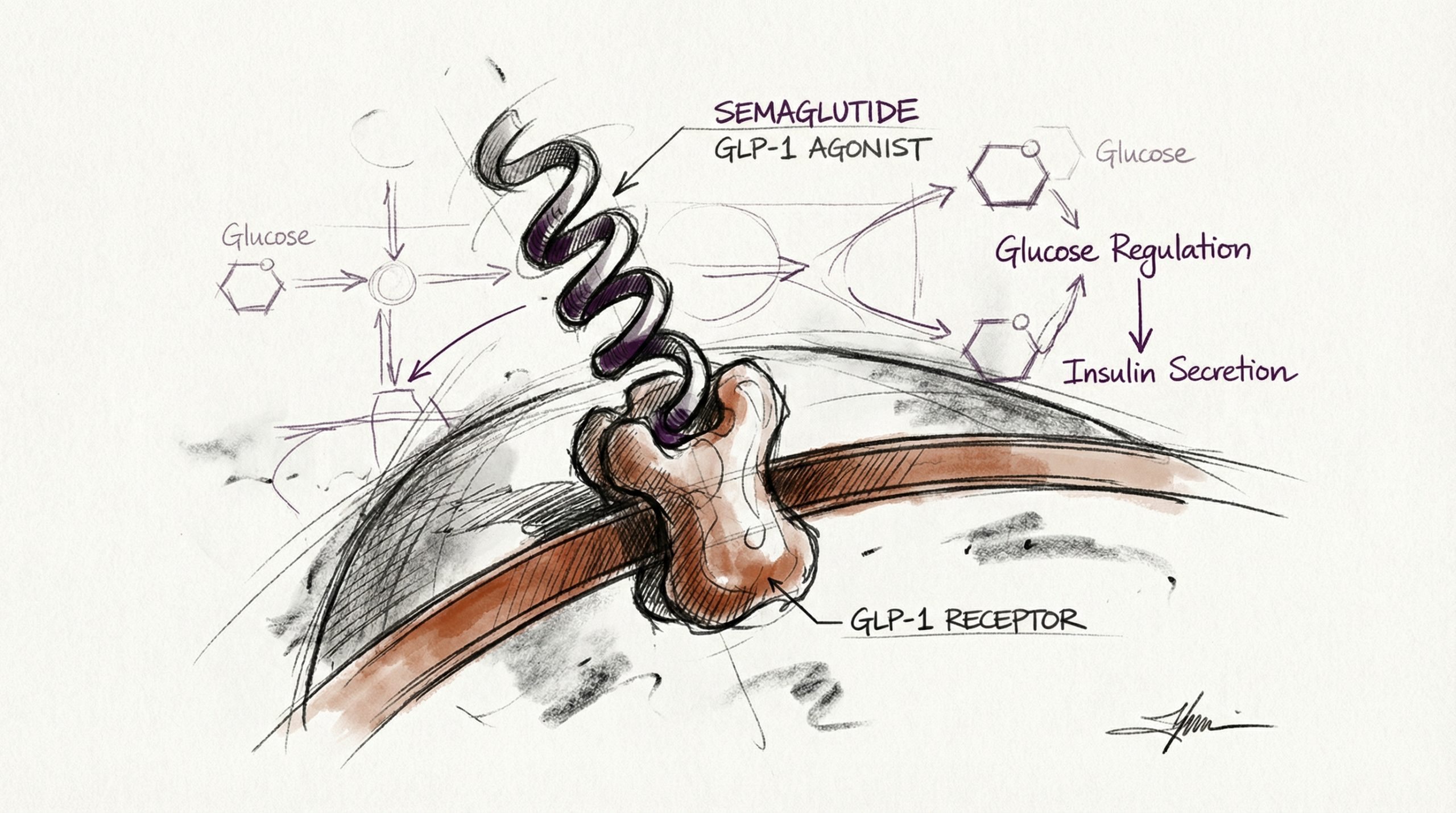
Semaglutide activates the GLP-1 receptor, a G-protein-coupled receptor expressed in pancreatic beta cells, the gastrointestinal tract, the central nervous system, and cardiovascular tissue. GLP-1 receptor activation triggers multiple downstream signaling cascades that collectively influence glucose metabolism, appetite, and energy balance. In pancreatic beta cells, GLP-1 receptor stimulation enhances glucose-dependent insulin secretion — meaning insulin is released only when blood glucose is elevated, reducing the risk of hypoglycemia observed with non-incretin-based compounds (PubMed: 28930514).
Beyond the pancreas, semaglutide’s effects on appetite regulation are mediated through GLP-1 receptors in the hypothalamus and brainstem. Preclinical research indicates that GLP-1 receptor activation in these regions reduces hunger signaling, increases satiety, and decreases food reward processing. Functional MRI studies in human subjects have demonstrated that semaglutide reduces neural responses to food cues in brain regions associated with appetite and reward (PubMed: 35441470). This central appetite suppression mechanism is considered the primary driver of the body weight reductions observed in clinical trials.
Semaglutide also slows gastric emptying, which contributes to prolonged satiety after meals. This effect is mediated through vagal nerve signaling and direct GLP-1 receptor activation in the gastrointestinal tract. The combination of central appetite suppression and delayed gastric emptying creates a dual mechanism for reducing caloric intake without requiring conscious dietary restriction.
What Do the Published Clinical Studies Show?
Semaglutide has one of the most robust clinical trial programs of any peptide compound, with multiple Phase III randomized controlled trials completed and published. The STEP (Semaglutide Treatment Effect in People with Obesity) trial program is the most comprehensive body of evidence for semaglutide’s effects on body weight.
The STEP 1 trial enrolled 1,961 adults with obesity (BMI ≥30) or overweight (BMI ≥27) with at least one weight-related comorbidity. Participants receiving semaglutide 2.4 mg weekly achieved a mean body weight reduction of 14.9% over 68 weeks, compared to 2.4% with placebo. More than one-third of participants (32%) achieved ≥20% weight reduction (PubMed: 33567185). These results were unprecedented for a pharmacological intervention and generated significant interest in GLP-1 agonist research.
The STEP 2 trial specifically examined participants with type 2 diabetes, reporting a mean weight reduction of 9.6% with semaglutide 2.4 mg versus 3.4% with placebo over 68 weeks (PubMed: 33667417). The SUSTAIN trial program examined glycemic outcomes, demonstrating significant HbA1c reductions compared to both placebo and active comparators including other injectable diabetes therapies.
Cardiovascular outcomes data from the SELECT trial demonstrated that semaglutide reduced major adverse cardiovascular events (MACE) by 20% in participants with established cardiovascular disease and obesity but without diabetes (PubMed: 37952131). This finding expanded the research interest in semaglutide beyond metabolic endpoints into cardiovascular protection.
How Does the GLP-1 Receptor System Work?
The GLP-1 receptor system is part of the incretin axis — a hormonal feedback loop between the gut and the pancreas that regulates postprandial glucose metabolism. When food enters the small intestine, enteroendocrine L-cells release native GLP-1 into the circulation. This GLP-1 travels to pancreatic beta cells and stimulates insulin secretion in a glucose-dependent manner. However, native GLP-1 is rapidly degraded by the enzyme dipeptidyl peptidase-4 (DPP-4), giving it a plasma half-life of only 2-3 minutes.
Semaglutide overcomes this limitation through two key structural modifications: an amino acid substitution at position 8 (Aib for Ala) that confers DPP-4 resistance, and a C-18 fatty diacid chain attached via a linker at position 26 that enables non-covalent binding to serum albumin. Albumin binding shields semaglutide from renal clearance and creates a circulating reservoir that maintains therapeutic concentrations for approximately 7 days. This extended half-life enables once-weekly administration in research protocols, a significant practical advantage over short-acting GLP-1 analogs that require daily or twice-daily administration.
What Are the Key Observations in Research Models?
Beyond the landmark clinical trials, published research has documented several additional observations with semaglutide. In preclinical models, GLP-1 receptor agonism has been associated with reduced hepatic lipid accumulation (steatosis), suggesting potential applications in non-alcoholic fatty liver disease (NAFLD) research (PubMed: 33110029). Renal protective effects have also been observed, with data suggesting GLP-1 receptor activation may reduce albuminuria and slow progression of chronic kidney disease markers in diabetic models.
Neurological research represents an emerging area of interest. Preclinical studies have investigated GLP-1 receptor agonists for neuroprotective properties, with some rodent models showing reduced neuroinflammation and improved cognitive markers. Early-stage clinical trials are exploring semaglutide in neurodegenerative conditions, though results are preliminary. Anti-inflammatory effects have been documented across multiple tissue types, with published data suggesting GLP-1 receptor activation reduces circulating inflammatory markers including C-reactive protein (CRP) and interleukin-6 (IL-6).
What Is the Standard Reconstitution Protocol for Semaglutide?
Research-grade semaglutide is supplied as a lyophilized powder, typically in 3 mg vials. Reconstitution follows standard peptide preparation protocols using bacteriostatic water (BAC water). Researchers typically reconstitute a 3 mg vial with 2 mL of BAC water, yielding a concentration of 1,500 mcg/mL. At this concentration, each unit on a 100-unit insulin syringe equals 15 mcg of semaglutide.
The reconstitution procedure follows the same protocol as other peptides: swab both vial stoppers with alcohol, inject BAC water slowly along the vial wall, allow 2-3 minutes for dissolution, and gently roll to mix. The reconstituted solution should be stored at 2-8 °C (36-46 °F) and used within 21-28 days. For detailed reconstitution instructions, see our Peptide Reconstitution 101 guide, or calculate exact syringe units with the reconstitution calculator at HowToMixPeptides.com.
What Research Dosages Are Referenced in Published Protocols?
Published research protocols for semaglutide use a titration approach, starting at lower dosages and increasing over several weeks. Clinical trials typically initiated at 250 mcg (0.25 mg) weekly, escalating in 4-week intervals to the target research dosage. The STEP trials used a final dosage of 2.4 mg weekly, while the SUSTAIN glycemic control trials primarily studied 0.5 mg and 1.0 mg weekly dosages.
For standardized laboratory protocols, a common reference dosage is 250 mcg (0.25 mg) administered once weekly. With a 3 mg vial reconstituted in 2 mL of BAC water (1,500 mcg/mL), this corresponds to approximately 17 units on a 100-unit insulin syringe. The once-weekly administration schedule reflects semaglutide’s ~7-day half-life. Research cycles of 8 weeks on followed by 8 weeks off are commonly referenced, though clinical trials have studied continuous administration for up to 104 weeks. Researchers comparing semaglutide to other metabolic peptides may find our semaglutide vs tirzepatide comparison useful for protocol design.
What Are the Documented Observations in Research Models?
The most commonly documented observations in published semaglutide research include gastrointestinal effects, particularly during the initial titration period. Published studies report nausea, reduced appetite, and altered bowel patterns as the most frequently observed changes in research subjects. The STEP trials documented nausea in approximately 44% of semaglutide participants versus 17% with placebo, though these observations typically diminished with continued administration and were the primary reason for the gradual titration protocol (PubMed: 33567185).
Published studies have also documented changes in body composition beyond total weight reduction. Dual-energy X-ray absorptiometry (DXA) data from clinical trials demonstrated that approximately 40% of weight loss with semaglutide was lean mass, prompting research interest in combining GLP-1 agonist protocols with resistance exercise or other interventions to preserve lean tissue. Researchers studying semaglutide should review the complete published literature on body composition changes when designing research protocols. For peptide-specific reconstitution data, see the peptide reference library at HowToMixPeptides.com.
What Are the Current Research Limitations?
Despite the robust clinical trial data, several important research questions remain unresolved. Long-term durability of weight reduction after discontinuation is a key concern — the STEP 1 extension study showed significant weight regain after semaglutide was stopped, with participants regaining approximately two-thirds of their lost weight within one year of cessation (PubMed: 35441470). This finding has generated interest in optimal cycling and maintenance protocols.
The lean mass loss component of semaglutide-associated weight reduction is an active area of investigation. Strategies to mitigate lean mass loss — including concurrent exercise protocols and protein intake optimization — are being studied but lack definitive published guidelines. Additionally, the long-term cardiovascular and metabolic effects of sustained GLP-1 receptor agonism beyond 2-3 years are not yet fully characterized, though the SELECT trial data is encouraging for cardiovascular safety.
Frequently Asked Questions
What is semaglutide?
Semaglutide is a synthetic GLP-1 receptor agonist — a modified 31-amino-acid peptide with a C-18 fatty acid chain that extends its half-life to approximately 7 days. It activates GLP-1 receptors in the pancreas, brain, and GI tract to influence glucose metabolism, appetite, and energy balance.
How is semaglutide reconstituted for research?
A 3 mg vial is typically reconstituted with 2 mL of bacteriostatic water, yielding 1,500 mcg/mL. BAC water is injected slowly along the vial wall, allowed to dissolve for 2-3 minutes, then gently rolled. Store at 2-8 °C (36-46 °F) and use within 21-28 days.
Why is semaglutide administered weekly instead of daily?
Semaglutide’s C-18 fatty acid modification enables albumin binding, which shields it from renal clearance and DPP-4 degradation. This gives it a plasma half-life of ~7 days, compared to 2-3 minutes for native GLP-1, making once-weekly administration sufficient to maintain research concentrations.
How does semaglutide compare to tirzepatide?
Semaglutide is a single-target GLP-1 receptor agonist, while tirzepatide is a dual GIP/GLP-1 receptor agonist. Published studies suggest tirzepatide may produce greater weight reduction at comparable dosages, likely due to additive effects from GIP receptor activation. See our detailed comparison.
What weight reduction has been observed in clinical trials?
The STEP 1 trial reported mean weight reduction of 14.9% over 68 weeks with semaglutide 2.4 mg weekly versus 2.4% with placebo. Over one-third of participants achieved 20% or greater weight reduction.
Does weight return after discontinuation?
Published extension data shows significant weight regain after semaglutide cessation — approximately two-thirds of lost weight was regained within one year of stopping. This has generated research interest in maintenance protocols and optimal cycling strategies.
What are the most commonly documented observations?
Gastrointestinal effects (nausea, reduced appetite, altered bowel patterns) are the most frequently reported observations, particularly during initial titration. The STEP trials documented nausea in 44% of semaglutide participants versus 17% with placebo, with observations typically diminishing over time.
Where can I find semaglutide with third-party purity verification?
Peptideware provides research-grade semaglutide with independent third-party HPLC and mass spectrometry verification. Certificates of analysis are published on the product page.
For research purposes only. All products and information are provided for laboratory and research purposes only.