Last Updated: March 2026 | v1.0
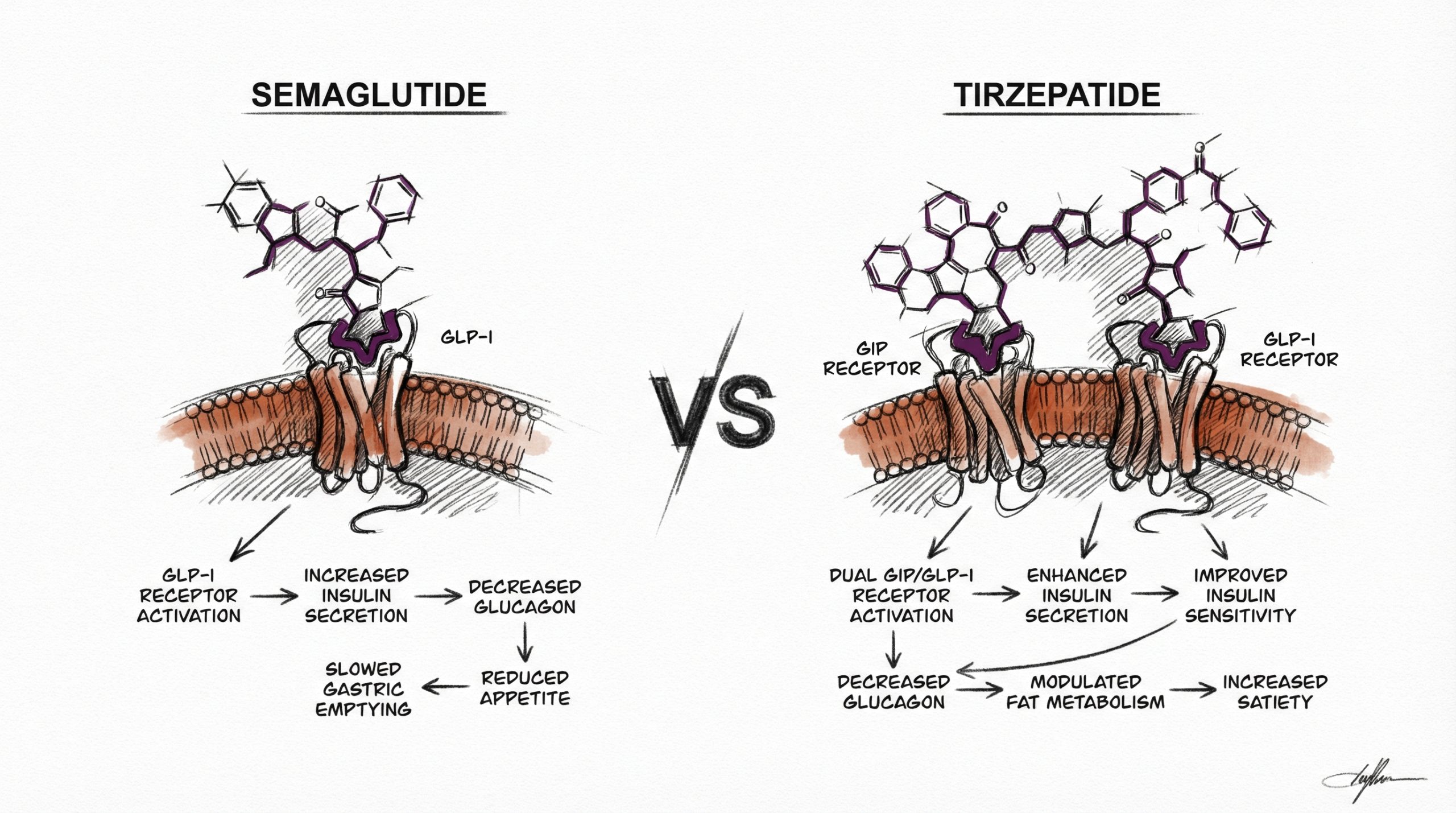
Semaglutide and tirzepatide are the two most studied incretin-based peptides in metabolic research. Both compounds target the GLP-1 receptor, but tirzepatide adds a second target — the glucose-dependent insulinotropic polypeptide (GIP) receptor — creating a dual agonist with distinct pharmacological properties. Head-to-head clinical trial data now exists comparing these compounds directly, giving researchers quantitative benchmarks for efficacy, body weight effects, and glycemic outcomes. Understanding the mechanistic differences between single-target GLP-1 agonism (semaglutide) and dual GIP/GLP-1 agonism (tirzepatide) is essential for designing informed metabolic research protocols. This guide compares both compounds across mechanism of action, published clinical data, reconstitution protocols, and research dosage parameters. All products and information are provided for laboratory and research purposes only.
Quick Comparison: Semaglutide vs Tirzepatide
- Semaglutide: GLP-1 receptor agonist, ~4,113 Da, ~7-day half-life, once-weekly dosing
- Tirzepatide: Dual GIP/GLP-1 receptor agonist, ~4,813 Da, ~5-day half-life, once-weekly or 3x/week dosing
- Weight reduction: STEP 1 (semaglutide): 14.9% mean reduction; SURMOUNT-1 (tirzepatide): 20.9% at highest dosage
- Key difference: Tirzepatide’s GIP receptor activation adds a second metabolic pathway not targeted by semaglutide
- Direct comparison: SURPASS-2 showed tirzepatide 15 mg achieved greater HbA1c reduction than semaglutide 1 mg
Use the free reconstitution calculator for exact syringe units, or compare protocols in the peptide reference library.
How Do the Mechanisms of Action Differ?
Semaglutide is a selective GLP-1 receptor agonist. It activates GLP-1 receptors in pancreatic beta cells (enhancing glucose-dependent insulin secretion), the hypothalamus and brainstem (reducing appetite and food reward signaling), and the gastrointestinal tract (slowing gastric emptying). Its structural modifications — an Aib substitution at position 8 and a C-18 fatty acid chain at position 26 — confer DPP-4 resistance and albumin binding, producing a plasma half-life of approximately 7 days. For a complete mechanism review, see our Semaglutide Research Guide.
Tirzepatide is a dual GIP/GLP-1 receptor agonist — the first approved compound in this class. Its 39-amino-acid sequence activates both the GIP receptor and the GLP-1 receptor, with approximately 5-fold greater affinity for GIP receptors relative to GLP-1 receptors. GIP (glucose-dependent insulinotropic polypeptide) is the other major incretin hormone, released by K-cells in the upper small intestine. GIP receptor activation enhances insulin secretion through a pathway complementary to but distinct from GLP-1, and emerging research suggests GIP signaling may also influence adipose tissue metabolism, lipid storage, and energy expenditure (PubMed: 35143424). The additive effects of dual receptor activation are the leading explanation for tirzepatide’s greater observed efficacy in weight reduction trials.
What Do the Head-to-Head Clinical Studies Show?
The SURPASS-2 trial provides the most direct published comparison. This randomized controlled trial compared tirzepatide (5 mg, 10 mg, and 15 mg) to semaglutide 1 mg in participants with type 2 diabetes. All three tirzepatide dosages achieved greater HbA1c reductions than semaglutide 1 mg: tirzepatide 15 mg reduced HbA1c by 2.30% versus 1.86% for semaglutide. Weight reduction was also greater with tirzepatide: -11.2 kg (tirzepatide 15 mg) versus -5.7 kg (semaglutide 1 mg) over 40 weeks (PubMed: 34170647).
The weight reduction data from separate trials also favors tirzepatide at the highest dosages. The SURMOUNT-1 trial (tirzepatide in participants with obesity without diabetes) reported mean weight reduction of 20.9% with tirzepatide 15 mg over 72 weeks (PubMed: 35658024), compared to 14.9% with semaglutide 2.4 mg in STEP 1 over 68 weeks. While cross-trial comparisons have methodological limitations (different populations, timeframes, and endpoints), the magnitude of difference is substantial and consistent with the dual-agonist mechanism.
How Do Reconstitution Protocols Compare?
Both peptides follow standard reconstitution procedures using bacteriostatic water (BAC water), but their vial sizes and dosing schedules differ.
Semaglutide reconstitution: A 3 mg vial reconstituted with 2 mL of BAC water yields 1,500 mcg/mL. For a research dosage of 250 mcg weekly, researchers draw approximately 17 units on a 100-unit insulin syringe. Administered once weekly, reflecting the ~7-day half-life.
Tirzepatide reconstitution: A 10 mg vial reconstituted with 2 mL of BAC water yields 5,000 mcg/mL. For a research dosage of 0.5 mg (500 mcg), researchers draw 10 units on a 100-unit insulin syringe. Published protocols reference administration 3 times per week, reflecting the ~5-day half-life.
Both compounds should be reconstituted by injecting BAC water slowly along the vial wall, allowing 2-3 minutes for dissolution, and gently rolling — never shaking. Store reconstituted solutions at 2-8 °C (36-46 °F) and use within 21-28 days. For exact unit calculations, use the reconstitution calculator at HowToMixPeptides.com.
How Do Research Dosages and Protocols Compare?
| Parameter | Semaglutide | Tirzepatide |
|---|---|---|
| Vial size | 3 mg | 10 mg |
| BAC water volume | 2 mL | 2 mL |
| Research dosage | 250 mcg | 500 mcg (0.5 mg) |
| Syringe units | 17 units | 10 units |
| Frequency | 1x/week | 3x/week |
| Half-life | ~7 days | ~5 days |
| Cycle | 8 weeks on / 8 weeks off | 8 weeks on / 8 weeks off |
The most significant protocol difference is administration frequency: semaglutide’s longer half-life enables once-weekly dosing, while tirzepatide is commonly referenced at three times per week in research protocols. Clinical trials used weekly dosing for both compounds, but research-grade tirzepatide protocols sometimes use more frequent administration at lower per-dose amounts.
Which Compound Shows Greater Efficacy in Published Data?
Based on published clinical trial data, tirzepatide at its highest studied dosages (15 mg weekly) produced greater reductions in both body weight and HbA1c compared to semaglutide at its highest studied dosages. The SURMOUNT-1 data showing 20.9% mean weight reduction with tirzepatide 15 mg exceeded the 14.9% reported for semaglutide 2.4 mg in STEP 1. In the direct head-to-head SURPASS-2 trial, tirzepatide 15 mg achieved approximately twice the weight reduction of semaglutide 1 mg.
However, direct comparisons require important caveats. The SURPASS-2 trial compared tirzepatide to semaglutide at 1 mg, not the 2.4 mg obesity dosage. Trial populations, durations, and endpoints differed. Semaglutide has substantially more long-term safety data from completed trials. Additionally, individual response variability is significant in both compound classes, and some research subjects may respond better to one mechanism than the other. Both compounds represent significant advances in metabolic peptide research, and the choice between them in research protocols should be guided by the specific research question being investigated.
What Observations Are Documented for Each Compound?
Both compounds share a similar observation profile dominated by gastrointestinal effects, consistent with their shared GLP-1 receptor agonism. Nausea, reduced appetite, and altered bowel patterns are the most frequently documented observations in clinical trials of both compounds. The SURPASS-2 trial reported comparable rates of gastrointestinal observations between tirzepatide and semaglutide, suggesting the GIP receptor component does not significantly add to GI-related effects.
Both compounds have been associated with lean mass loss as a component of total weight reduction — approximately 25-40% of weight lost is lean mass. This observation has generated research interest in combining incretin-based protocols with resistance exercise and protein optimization strategies. Cardiovascular safety has been demonstrated for semaglutide (SELECT trial) and is being investigated for tirzepatide in ongoing trials. Researchers can source both semaglutide and tirzepatide from Peptideware with third-party purity verification.
Frequently Asked Questions
What is the main difference between semaglutide and tirzepatide?
Semaglutide targets only the GLP-1 receptor, while tirzepatide targets both the GIP and GLP-1 receptors. This dual mechanism provides additive metabolic effects that published data suggests produces greater weight reduction and glycemic improvement at comparable dosages.
Which compound produces more weight reduction in clinical trials?
Tirzepatide at its highest dosage (15 mg weekly) produced 20.9% mean weight reduction in SURMOUNT-1, compared to 14.9% with semaglutide 2.4 mg in STEP 1. In the direct SURPASS-2 comparison, tirzepatide 15 mg approximately doubled the weight reduction of semaglutide 1 mg.
Can semaglutide and tirzepatide be used together?
No published protocols describe concurrent use of both compounds. Since both activate the GLP-1 receptor, combining them would create overlapping receptor activation without clear additive benefit. Researchers typically study one compound per protocol.
Which has a longer half-life?
Semaglutide has a longer half-life (~7 days) compared to tirzepatide (~5 days). This is why semaglutide research protocols typically use once-weekly administration, while tirzepatide may be administered more frequently in some research protocols.
Which has more published safety data?
Semaglutide has substantially more long-term published safety data, including the completed SELECT cardiovascular outcomes trial. Tirzepatide’s long-term cardiovascular safety is being evaluated in ongoing trials.
Are the gastrointestinal observations different?
Both compounds produce similar gastrointestinal observations (nausea, reduced appetite, altered bowel patterns) due to shared GLP-1 receptor agonism. The SURPASS-2 head-to-head trial reported comparable GI observation rates between the two compounds.
For research purposes only. All products and information are provided for laboratory and research purposes only.