Last Updated: March 2026 | v1.0
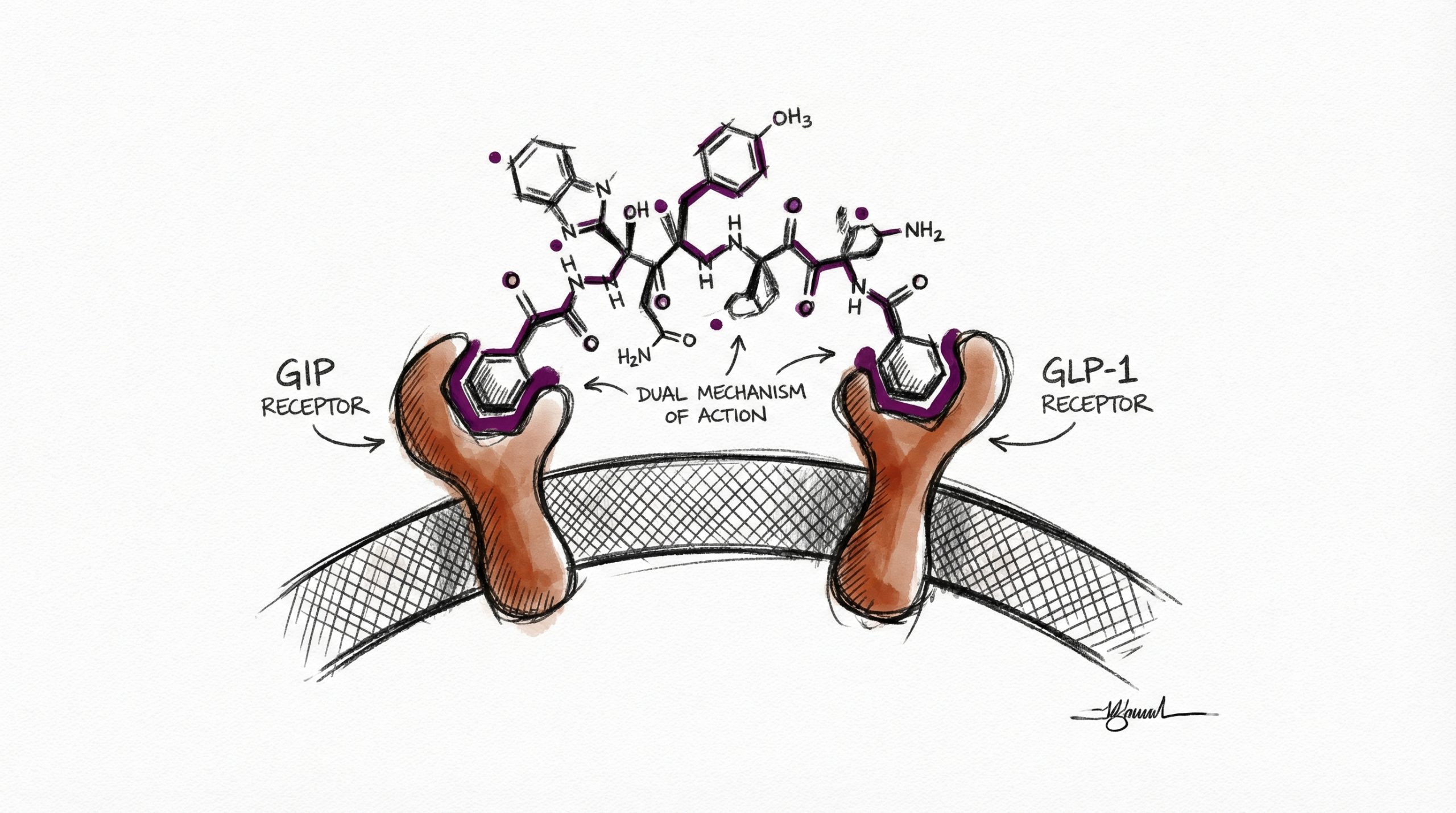
Tirzepatide is a synthetic 39-amino-acid peptide that functions as a dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist — the first compound approved in this dual-agonist class. With a molecular weight of approximately 4,813 Da, tirzepatide was engineered to activate both incretin receptors simultaneously, producing additive metabolic effects that exceed those observed with single-target GLP-1 agonists. Published data from the SURMOUNT and SURPASS clinical trial programs demonstrate unprecedented body weight reductions and glycemic improvements, establishing tirzepatide as one of the most studied metabolic peptides in clinical research. The compound’s C-20 fatty acid modification enables albumin binding and a plasma half-life of approximately 5 days, supporting once-weekly or multiple-times-weekly dosing in research protocols. This guide covers tirzepatide’s dual-agonist mechanism, key clinical trial results, and standard laboratory protocols. All products and information are provided for laboratory and research purposes only.
Quick Facts: Tirzepatide
- Classification: Dual GIP/GLP-1 receptor agonist (first in class)
- Molecular weight: ~4,813 Da
- Half-life: ~5 days
- Receptor affinity: ~5x greater affinity for GIP receptor than GLP-1 receptor
- Primary research areas: Body weight regulation, glycemic control, energy metabolism, cardiovascular outcomes
Use the free reconstitution calculator for exact syringe units, or browse the tirzepatide reference page for dosing and protocol data.
What Is the Mechanism of Action of Tirzepatide?
Tirzepatide’s mechanism involves simultaneous activation of two G-protein-coupled receptors in the incretin system: the GIP receptor and the GLP-1 receptor. This dual activation distinguishes it from single-target GLP-1 agonists like semaglutide and liraglutide.
GLP-1 receptor activation produces effects well-characterized from single-agonist research: glucose-dependent insulin secretion from pancreatic beta cells, appetite suppression via hypothalamic GLP-1 receptors, and delayed gastric emptying through vagal nerve signaling. These effects reduce food intake and improve postprandial glucose regulation.
GIP receptor activation adds a second layer of metabolic effects. GIP (glucose-dependent insulinotropic polypeptide) is the dominant incretin hormone, responsible for approximately 60-70% of the postprandial insulin response in healthy individuals. GIP receptor activation enhances insulin secretion through a pathway complementary to GLP-1 signaling. Emerging research suggests GIP receptor agonism also influences adipose tissue directly — promoting lipid storage in subcutaneous fat (the metabolically favorable depot) rather than visceral fat, and potentially enhancing energy expenditure through brown adipose tissue activation (PubMed: 35143424).
Tirzepatide’s peptide backbone is based on the native GIP sequence with modifications that confer GLP-1 receptor cross-reactivity. A C-20 fatty diacid attached via a linker at position 20 enables non-covalent albumin binding, extending the plasma half-life to approximately 5 days and reducing renal clearance. Tirzepatide has approximately 5-fold greater affinity for the GIP receptor relative to the GLP-1 receptor, which may contribute to its differentiated metabolic profile compared to pure GLP-1 agonists.
What Do the SURMOUNT Clinical Trials Show?
The SURMOUNT trial program examined tirzepatide for body weight reduction in participants with obesity. SURMOUNT-1, the pivotal trial, enrolled 2,539 adults with BMI ≥30 (or ≥27 with comorbidities) without diabetes. The results were unprecedented for any pharmacological intervention:
At the 15 mg dosage over 72 weeks, tirzepatide produced a mean body weight reduction of 20.9% (approximately 24 kg / 52 lbs). Over half of participants (56.7%) achieved ≥20% weight reduction, and 36.2% achieved ≥25% weight reduction (PubMed: 35658024). These percentages exceeded any previously published pharmacological weight reduction data and approached the range historically associated only with bariatric surgical procedures.
SURMOUNT-2 examined tirzepatide specifically in participants with type 2 diabetes and obesity, reporting mean weight reductions of 12.8% (10 mg) and 14.7% (15 mg) over 72 weeks — significant reductions even in a population where weight loss is typically more challenging due to insulin resistance and diabetes medications that promote weight gain (PubMed: 37385275).
What Do the SURPASS Clinical Trials Show?
The SURPASS trial program focused on glycemic control in type 2 diabetes. SURPASS-2 is particularly significant because it directly compared tirzepatide to semaglutide 1 mg. All three tirzepatide dosages (5 mg, 10 mg, 15 mg) achieved greater HbA1c reductions than semaglutide 1 mg over 40 weeks. Tirzepatide 15 mg reduced HbA1c by 2.30% versus 1.86% for semaglutide. Weight reduction was also significantly greater: -11.2 kg with tirzepatide 15 mg versus -5.7 kg with semaglutide 1 mg (PubMed: 34170647).
SURPASS-1 through SURPASS-5 collectively demonstrated consistent HbA1c reductions across different patient populations and comparators. In SURPASS-4, tirzepatide was compared to insulin glargine in participants with type 2 diabetes and high cardiovascular risk, showing superior glycemic control and significant weight reduction compared to weight gain with insulin. These head-to-head comparisons against established therapies reinforced tirzepatide’s position as a leading research compound in metabolic science.
What Role Does GIP Play Beyond GLP-1?
The GIP receptor component of tirzepatide’s mechanism is an active area of research that distinguishes it from pure GLP-1 agonists. Traditional understanding viewed GIP primarily as an insulin secretagogue, but emerging research reveals broader metabolic functions. In adipose tissue, GIP receptor activation appears to promote efficient lipid storage and may enhance the thermogenic capacity of brown adipose tissue. In bone, GIP signaling has been linked to reduced bone resorption — a potentially favorable effect given concerns about bone density loss with significant weight reduction.
Preclinical research indicates that GIP receptor agonism may also attenuate the nausea associated with GLP-1 receptor activation. In animal models, combined GIP/GLP-1 receptor activation produced less nausea-related behavior than GLP-1 activation alone at equipotent doses. If confirmed in human studies, this could explain why tirzepatide produces comparable or greater efficacy with a tolerability profile similar to pure GLP-1 agonists despite stronger overall receptor activation (PubMed: 35143424).
What Is the Standard Reconstitution Protocol for Tirzepatide?
Research-grade tirzepatide is supplied as a lyophilized powder, typically in 10 mg vials. Researchers typically reconstitute with 2 mL of bacteriostatic water (BAC water), producing a concentration of 5,000 mcg/mL. At this concentration, each unit on a 100-unit insulin syringe equals 50 mcg of tirzepatide.
Reconstitution follows standard peptide protocols: swab both vial stoppers with alcohol, inject BAC water slowly along the vial wall, allow 2-3 minutes for dissolution, and gently roll to mix. Store the reconstituted solution at 2-8 °C (36-46 °F) and use within 21-28 days. For step-by-step reconstitution instructions, see our Peptide Reconstitution 101 guide, or use the reconstitution calculator at HowToMixPeptides.com.
What Research Dosages Are Referenced in Published Protocols?
Clinical trials used dosages of 5 mg, 10 mg, and 15 mg administered once weekly via subcutaneous injection. All trials employed a dose-escalation protocol, starting at 2.5 mg weekly and increasing by 2.5 mg every 4 weeks to the target dosage. This gradual titration minimizes gastrointestinal observations during the initial adjustment period.
For standardized laboratory protocols, a commonly referenced research dosage is 0.5 mg (500 mcg) administered subcutaneously 3 times per week. With a 10 mg vial reconstituted in 2 mL of BAC water (5,000 mcg/mL), this corresponds to 10 units on a 100-unit insulin syringe per administration. Research cycles of 8 weeks on followed by 8 weeks off are commonly referenced. Researchers comparing tirzepatide to semaglutide should review our semaglutide vs tirzepatide comparison for protocol design. Additional peptide references are available at HowToMixPeptides.com.
What Observations Are Documented in Clinical Trials?
The observation profile of tirzepatide is dominated by gastrointestinal effects, consistent with its GLP-1 receptor agonist activity. In the SURMOUNT-1 trial, the most frequently reported observations were nausea (24-33% depending on dosage), diarrhea (17-23%), and constipation (11-17%). These observations were predominantly mild to moderate in severity and decreased over time, particularly after the initial dose-escalation period. Discontinuation due to gastrointestinal observations occurred in approximately 4-7% of tirzepatide participants.
Body composition analyses from the SURMOUNT trials showed that approximately 25-33% of total weight lost was lean mass — slightly lower than the ~40% lean mass loss reported with semaglutide in some analyses. This difference, if confirmed in head-to-head body composition studies, could suggest that GIP receptor activation helps preserve lean tissue during caloric deficit, though this remains an area of active investigation. Researchers studying tirzepatide should review the complete published observation data when designing research protocols.
What Are the Current Research Limitations?
Despite the robust clinical trial data, several research questions remain open. Long-term cardiovascular outcomes data specific to tirzepatide is still being generated — the SURPASS-CVOT trial is ongoing. While semaglutide’s SELECT trial demonstrated a 20% MACE reduction, equivalent data for tirzepatide is pending. Long-term weight maintenance after tirzepatide cessation has shown results similar to semaglutide, with significant weight regain after discontinuation.
The relative contribution of GIP versus GLP-1 receptor activation to tirzepatide’s overall metabolic effects is not yet fully delineated. Understanding which benefits are driven by GIP agonism, which by GLP-1 agonism, and which require dual activation simultaneously would inform the development of next-generation compounds. Additionally, the optimal dosing strategy for research-grade tirzepatide (including frequency, titration approach, and cycle duration) has not been established through controlled studies outside of the clinical trial protocols.
Frequently Asked Questions
What is tirzepatide?
Tirzepatide is a synthetic 39-amino-acid dual GIP/GLP-1 receptor agonist — the first approved compound in this class. It activates both incretin receptors simultaneously, producing additive metabolic effects that exceed single-target GLP-1 agonists.
How does tirzepatide differ from semaglutide?
Semaglutide targets only the GLP-1 receptor. Tirzepatide targets both GIP and GLP-1 receptors, with ~5x greater GIP affinity. Published data shows tirzepatide produces greater weight reduction (20.9% vs 14.9%) and glycemic improvement at their respective highest dosages. See our detailed comparison.
What weight reduction has been observed with tirzepatide?
SURMOUNT-1 reported 20.9% mean weight reduction with tirzepatide 15 mg over 72 weeks. Over half of participants (56.7%) achieved 20% or greater weight reduction — results approaching those historically seen only with bariatric surgery.
How is tirzepatide reconstituted for research?
A 10 mg vial is typically reconstituted with 2 mL of bacteriostatic water, yielding 5,000 mcg/mL. For a research dosage of 500 mcg, draw 10 syringe units. Store at 2-8 °C (36-46 °F) and use within 21-28 days.
What is the GIP receptor and why does it matter?
GIP (glucose-dependent insulinotropic polypeptide) is the dominant incretin hormone, responsible for 60-70% of postprandial insulin response. GIP receptor activation enhances insulin secretion and may influence adipose tissue metabolism and energy expenditure beyond GLP-1 effects alone.
What are the most commonly observed effects in trials?
Gastrointestinal observations (nausea, diarrhea, constipation) are most common, predominantly mild-to-moderate, and diminish after dose escalation. Discontinuation rates due to these observations were 4-7% in SURMOUNT trials.
Where can I find tirzepatide with purity verification?
Peptideware provides research-grade tirzepatide with independent third-party HPLC and mass spectrometry verification. COAs are published on the product page.
For research purposes only. All products and information are provided for laboratory and research purposes only.