Last Updated: March 2026 | v1.0
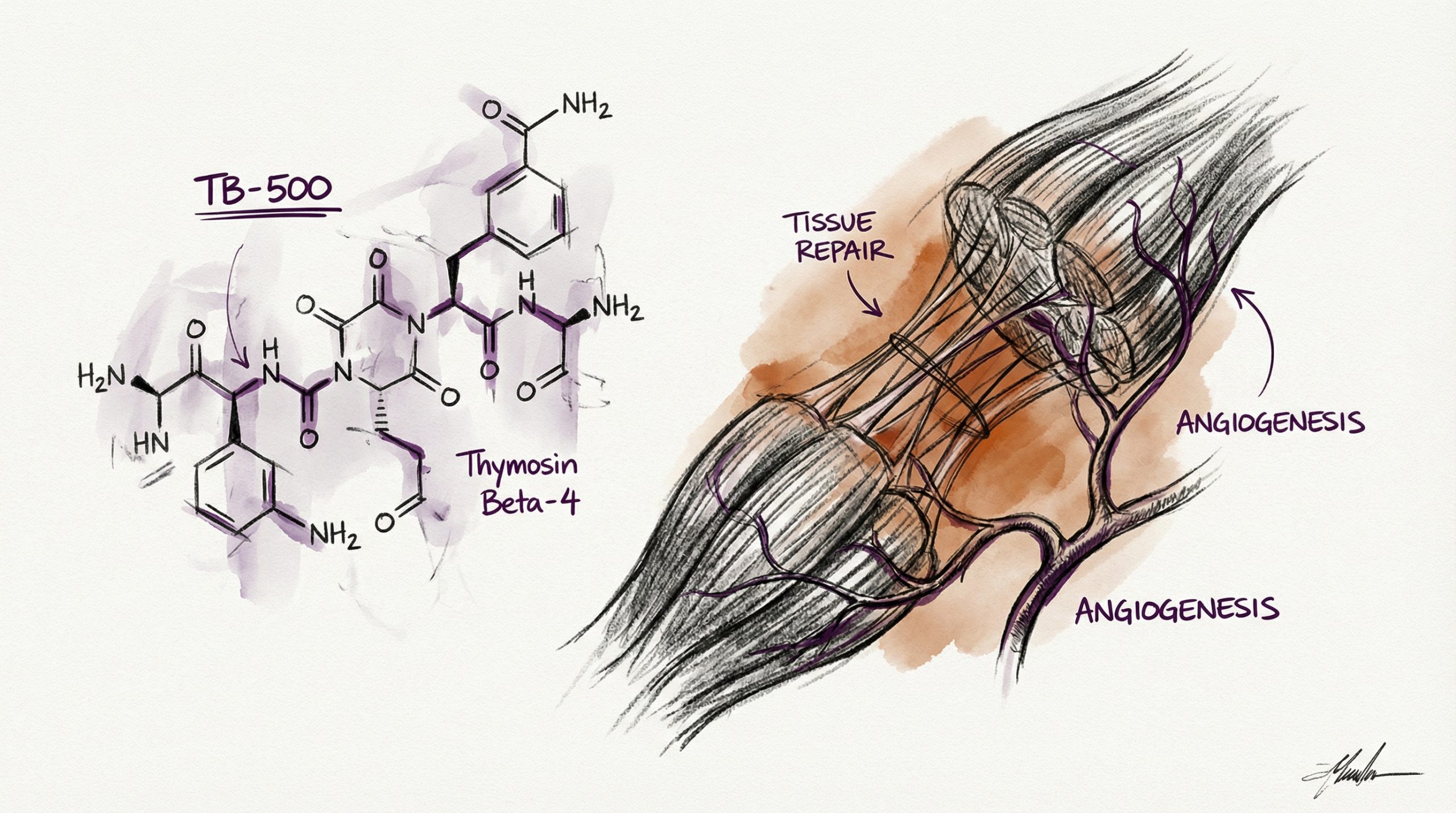
TB-500 is a synthetic 43-amino-acid peptide corresponding to the active region of thymosin beta-4 (Tβ4), a 4,963 Da naturally occurring protein found in virtually all human and animal cells. Thymosin beta-4 is the primary intracellular G-actin sequestering protein — it regulates the polymerization of actin, the structural protein that forms the cytoskeleton of every cell. By controlling actin dynamics, Tβ4 influences fundamental cellular processes including migration, proliferation, differentiation, and survival. TB-500 replicates the active domain of thymosin beta-4 and has been studied in preclinical models of wound healing, cardiac repair, corneal healing, hair growth, and inflammatory modulation. The compound’s ability to promote cell migration into damaged tissue makes it one of the most studied peptides in tissue repair research. This guide covers TB-500’s mechanism of action, published preclinical and clinical findings, and standard laboratory protocols. All products and information are provided for laboratory and research purposes only.
Quick Facts: TB-500
- Classification: Synthetic fragment of thymosin beta-4 (Tβ4), 43 amino acids
- Molecular weight: 4,963 Da
- Primary mechanism: G-actin sequestration — regulates actin polymerization and cell migration
- Key research areas: Wound healing, cardiac repair, corneal healing, hair follicle regeneration, anti-inflammatory effects
- Distinguishing property: Promotes cell migration to injury sites rather than building new vasculature
Use the free reconstitution calculator for exact syringe units, or browse the TB-500 reference page for dosing and protocol data.
What Is the Mechanism of Action of TB-500?
TB-500’s primary mechanism centers on the regulation of actin polymerization through G-actin sequestration. Actin exists in two forms inside cells: globular actin (G-actin, the monomer) and filamentous actin (F-actin, the polymer). F-actin filaments form the structural skeleton of cells and are essential for cell shape, motility, and division. TB-500 binds to G-actin monomers and prevents them from spontaneously polymerizing into F-actin. This might seem counterintuitive — why would inhibiting actin polymerization promote healing? The answer lies in cytoskeletal remodeling.
When cells need to migrate (as they do during wound healing), they must disassemble existing actin structures, reshape themselves, and extend in new directions. TB-500’s sequestration of G-actin creates a pool of readily available monomers that can be rapidly deployed when and where the cell needs them, while simultaneously preventing disorganized polymerization. Published studies suggest this mechanism enables faster, more directed cell migration into wound sites (PubMed: 20614940).
Beyond actin regulation, TB-500 has been shown to upregulate the expression of laminin-5 in dermal models — an extracellular matrix protein critical for cell adhesion and migration at wound borders (PubMed: 17318735). TB-500 also promotes the formation of new blood vessels, though through a mechanism distinct from BPC-157’s VEGF pathway. TB-500’s angiogenic effects are believed to be downstream of its cell migration promotion, as endothelial cells must migrate and organize to form new capillary structures.
What Does the Published Research Show for Wound Healing?
Dermal wound healing is one of TB-500’s most thoroughly studied applications. In a landmark study, Malinda et al. demonstrated that thymosin beta-4 accelerated wound closure in a full-thickness dermal punch wound model. Treated wounds showed increased keratinocyte migration, enhanced angiogenesis at the wound bed, and organized collagen deposition compared to controls (PubMed: 10229405). The researchers attributed the accelerated closure specifically to enhanced cell migration rather than increased proliferation — an important mechanistic distinction.
Follow-up studies confirmed these findings and extended them to chronic wound models. In diabetic mouse wound models (which mimic the impaired healing observed in diabetic conditions), thymosin beta-4 treatment significantly improved wound closure rates and tissue organization. The improvement was particularly notable because diabetic wounds are characterized by impaired cell migration — precisely the process TB-500 enhances (PubMed: 17318735). Corneal wound healing studies have shown similar results, with thymosin beta-4 promoting corneal epithelial cell migration and reducing inflammation after injury.
What Has Been Studied in Cardiac Repair Research?
Cardiac tissue repair represents one of the most significant areas of TB-500/thymosin beta-4 research. In a pivotal 2004 study published in Nature, Bock-Marquette et al. demonstrated that thymosin beta-4 promoted cardiomyocyte survival after myocardial infarction in murine models. Treated animals showed preserved cardiac function, reduced infarct size, and enhanced formation of new cardiac blood vessels (PubMed: 15340520). This study was notable because adult cardiac muscle has extremely limited regenerative capacity, and thymosin beta-4 appeared to activate epicardial progenitor cells — dormant stem-like cells on the heart’s outer surface.
Subsequent research expanded on these findings. Studies showed that thymosin beta-4 pre-treatment (administered before cardiac injury) was particularly effective, suggesting a cardioprotective effect in addition to reparative function. The compound was shown to activate the Akt survival pathway in cardiomyocytes, reducing apoptosis (programmed cell death) in the peri-infarct zone. These cardiac findings generated sufficient interest that thymosin beta-4 was advanced into Phase I clinical trials for cardiac repair, though results of these trials are beyond the scope of this research compound review.
What Are the Anti-Inflammatory Research Findings?
Published research documents anti-inflammatory properties of TB-500 across multiple tissue types. In a corneal inflammation model, thymosin beta-4 reduced polymorphonuclear leukocyte (PMN) infiltration and decreased inflammatory cytokine expression (PubMed: 11856767). The anti-inflammatory mechanism appears to involve NF-κB pathway modulation — TB-500 has been shown to reduce NF-κB activation, which is a master regulator of inflammatory gene expression.
In joint tissue models, thymosin beta-4 reduced inflammatory markers and showed chondroprotective effects, leading to research interest in musculoskeletal inflammatory conditions. Hair follicle research has also documented anti-inflammatory effects, with thymosin beta-4 promoting the anagen (growth) phase of hair cycling while reducing perifollicular inflammation. These anti-inflammatory findings complement TB-500’s cell migration effects — reducing inflammation at wound sites creates a more favorable environment for the migrating repair cells that TB-500 promotes.
What Is the Standard Reconstitution Protocol for TB-500?
TB-500 for research use is supplied as a lyophilized powder, typically in 10 mg vials. Researchers typically reconstitute with 2 mL of bacteriostatic water (BAC water), producing a concentration of 5,000 mcg/mL. At this concentration, each unit on a 100-unit insulin syringe equals 50 mcg of TB-500.
The reconstitution procedure follows standard peptide protocols: swab both vial stoppers with 70% isopropyl alcohol, draw 2 mL BAC water into a sterile syringe, inject slowly along the inside wall of the peptide vial, allow 2-3 minutes for dissolution, and gently roll the vial to mix. The reconstituted solution should be clear and colorless. Store at 2-8 °C (36-46 °F) and use within 21-28 days. For step-by-step instructions, see our Peptide Reconstitution 101 guide, or use the reconstitution calculator at HowToMixPeptides.com.
What Research Dosages Are Referenced in Published Protocols?
Preclinical studies have used a wide range of thymosin beta-4 dosages depending on the animal model and route of administration. For standardized laboratory protocols, a commonly referenced research dosage is 500 mcg (0.5 mg) administered subcutaneously once daily (AM). With a 10 mg vial reconstituted in 2 mL of BAC water, this corresponds to 10 units on a 100-unit insulin syringe.
Published protocols typically describe daily administration for research cycles of 8 weeks on followed by 8 weeks off. Some protocols use a loading phase with higher initial dosages for the first 1-2 weeks before transitioning to maintenance dosing. Researchers studying TB-500 alongside BPC-157 should review our BPC-157 vs TB-500 comparison for protocol design considerations. For reconstitution data on additional peptides, visit the peptide reference library at HowToMixPeptides.com.
How Does TB-500 Compare to BPC-157?
TB-500 and BPC-157 are frequently studied together but operate through distinct mechanisms. TB-500 promotes tissue repair primarily through enhanced cell migration via actin regulation, while BPC-157 promotes repair primarily through angiogenesis via VEGF upregulation. In simplified terms: TB-500 moves repair cells to where they are needed, and BPC-157 builds the blood vessel infrastructure to supply those cells with oxygen and nutrients. This mechanistic complementarity is why many published protocols study both compounds concurrently. TB-500 is available from Peptideware with third-party purity verification. For a detailed comparison of both compounds, see our BPC-157 vs TB-500 research guide.
What Are the Current Research Limitations?
TB-500 research has several important limitations. The majority of published data comes from animal models, particularly rodent and equine studies. Thymosin beta-4 has been used extensively in veterinary contexts, particularly in equine sports medicine, but this data does not directly translate to other research applications. The Phase I cardiac repair trials referenced earlier represent the most advanced human studies, but comprehensive efficacy data from controlled clinical trials is limited.
Additionally, the optimal dosing parameters (frequency, duration, route of administration) for TB-500 have not been established through dose-finding clinical trials. Published preclinical protocols vary significantly in their dosing approaches. The relationship between TB-500’s in vitro effects on actin dynamics and its in vivo tissue repair effects is not fully characterized — the concentration of TB-500 reaching target tissues after subcutaneous administration may differ substantially from the concentrations used in cell culture studies.
Frequently Asked Questions
What is TB-500 derived from?
TB-500 is a synthetic 43-amino-acid peptide corresponding to the active region of thymosin beta-4 (Tβ4), a naturally occurring protein found in virtually all human and animal cells. It replicates the actin-binding domain responsible for Tβ4’s cell migration and tissue repair effects.
How does TB-500 promote tissue repair?
TB-500 sequesters G-actin monomers, which promotes cytoskeletal remodeling and enables directed cell migration into damaged tissue. It also upregulates laminin-5 for cell adhesion and promotes angiogenesis through endothelial cell migration.
What is the standard research dosage for TB-500?
A commonly referenced research dosage is 500 mcg administered subcutaneously once daily (AM). With a 10 mg vial reconstituted in 2 mL BAC water, this equals 10 syringe units. Protocols typically use 8-week on / 8-week off cycles.
Can TB-500 be combined with BPC-157?
Yes. Combined protocols are documented in published literature. The two peptides target complementary mechanisms — TB-500 promotes cell migration while BPC-157 promotes angiogenesis — providing broader tissue repair pathway coverage.
What makes TB-500 different from full-length thymosin beta-4?
TB-500 is a synthetic fragment containing the active actin-binding domain of the full 43-amino-acid thymosin beta-4 sequence. It replicates the key functional region responsible for cell migration and tissue repair activity.
How long does reconstituted TB-500 remain stable?
Reconstituted with BAC water and stored at 2-8 °C (36-46 °F), TB-500 remains stable for approximately 21-28 days. Lyophilized TB-500 stored at -20 °C (-4 °F) can remain viable for 12-24 months.
Where can I find TB-500 with third-party testing?
Peptideware provides TB-500 (10 mg) with independent HPLC and mass spectrometry verification. COAs are published on the product page.
For research purposes only. All products and information are provided for laboratory and research purposes only.