Last Updated: April 14, 2026
Mitochondrial research compounds sit at the intersection of bioenergetics, longevity biology, and cellular aging science. Among the most studied molecules in this space are NAD+ (nicotinamide adenine dinucleotide) and SS-31 (Szeto-Schiller peptide 31, also known as Elamipretide). Both are investigated in preclinical models for their role in supporting mitochondrial function, but they operate through fundamentally different mechanisms and physical chemistries.
An important honesty note up front: NAD+ is not technically a peptide. It is a dinucleotide coenzyme composed of two nucleotides joined through phosphate groups. SS-31, by contrast, is a synthetic tetrapeptide (D-Arg-2′,6′-Dmt-Lys-Phe-NH2) designed to localize inside mitochondria. Comparing them here makes scientific sense because both are researched for mitochondrial support, but they occupy very different molecular categories.
This comparison explores what published studies suggest about each molecule, how they target distinct mitochondrial compartments, and which research contexts favor one approach over the other. Research protocols are strictly for laboratory use; neither compound is intended for human consumption.
Quick Comparison Table
| Attribute | NAD+ | SS-31 (Elamipretide) |
|---|---|---|
| Molecular class | Dinucleotide coenzyme | Synthetic tetrapeptide |
| Molecular weight | ~663 Da | ~640 Da |
| Primary target location | Mitochondrial matrix / cytosol (ETC cofactor pool) | Inner mitochondrial membrane (cardiolipin domain) |
| Mechanism | Electron carrier; sirtuin and PARP substrate | Binds cardiolipin; stabilizes cristae architecture |
| Research format | 500mg lyophilized powder | 10mg lyophilized powder |
| Typical reconstitution | Bacteriostatic water for research | Bacteriostatic water for research |
| Research focus areas | Sirtuin pathway, NAD salvage, bioenergetic decline | Cardiolipin peroxidation, cristae morphology, ischemia-reperfusion models |
What Is Mitochondrial Research?
Mitochondria are double-membrane organelles that generate ATP through oxidative phosphorylation. They also regulate calcium signaling, apoptosis, and reactive oxygen species (ROS). Preclinical research indicates that mitochondrial dysfunction is a hallmark of biological aging, implicated in declining tissue bioenergetics, impaired proteostasis, and increased oxidative damage.
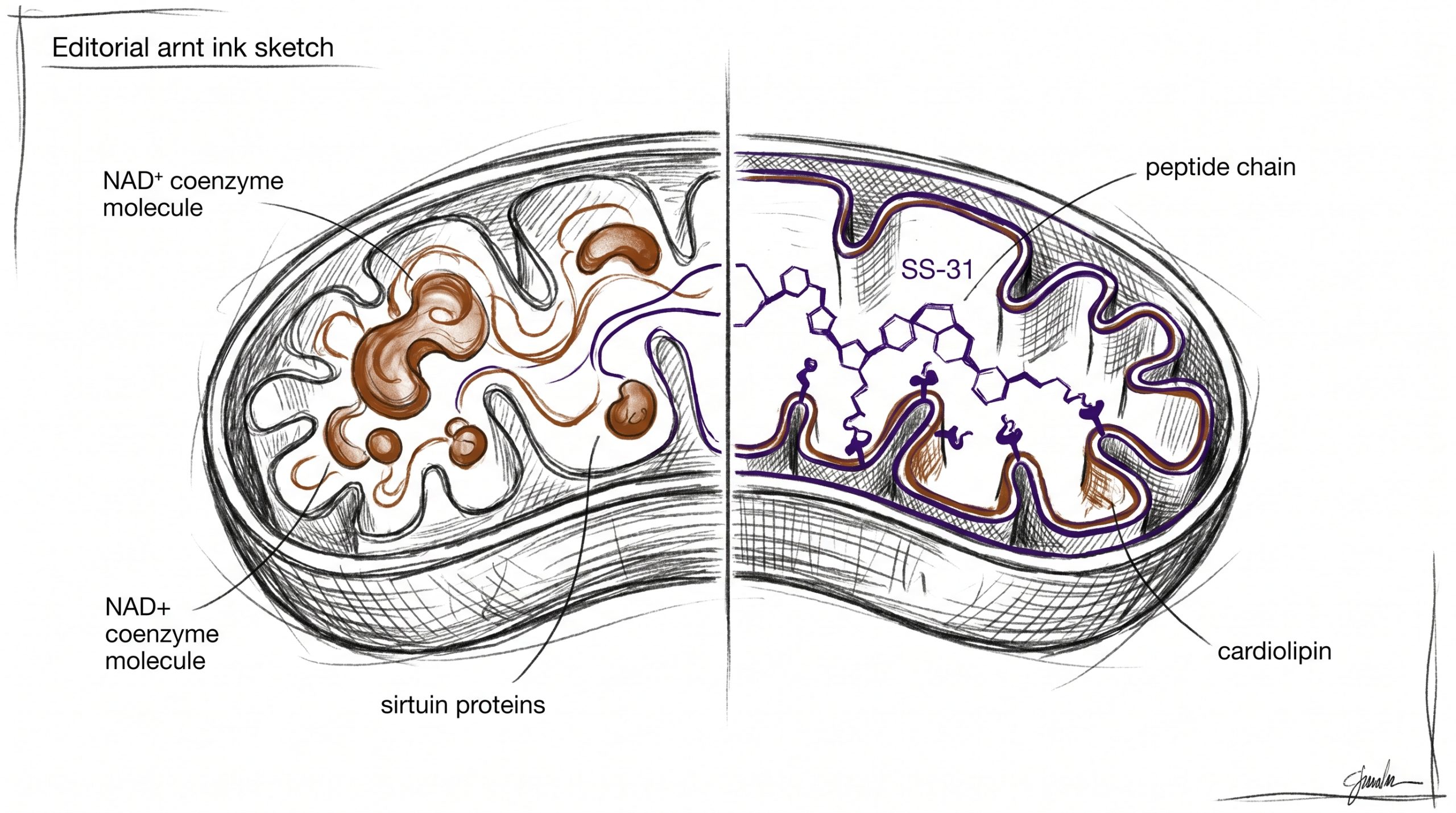
Two mitochondrial features drive much of the current research interest. First, the sirtuin pathway — a family of NAD+-dependent deacetylase enzymes (SIRT1-SIRT7) — regulates metabolic adaptation, DNA repair, and mitochondrial biogenesis. Sirtuin activity requires NAD+ as a co-substrate, and published studies suggest cellular NAD+ pools decline with age in multiple tissues.
Second, cardiolipin, a phospholipid concentrated in the inner mitochondrial membrane, organizes cristae architecture and anchors electron transport chain (ETC) complexes. Cardiolipin peroxidation disrupts supercomplex assembly and is observed in preclinical models of heart failure, ischemia-reperfusion injury, and neurodegeneration. Research compounds that preserve cardiolipin integrity are investigated for their ability to maintain mitochondrial membrane structure.
Understanding these two axes — the NAD+/sirtuin bioenergetic axis and the cardiolipin/cristae structural axis — is essential for interpreting how NAD+ and SS-31 differ.
How Does NAD+ Function?
NAD+ operates as a central electron carrier in cellular metabolism. In glycolysis, the TCA cycle, and beta-oxidation, NAD+ accepts electrons to become NADH, which then donates those electrons to Complex I of the electron transport chain. This redox cycling is the biochemical foundation of ATP production through oxidative phosphorylation.
Beyond its role as a cofactor, NAD+ is consumed as a substrate by three enzyme families: sirtuins (deacetylation), PARPs (poly-ADP-ribose polymerases, involved in DNA damage response), and CD38 (an ectoenzyme that degrades NAD+). Preclinical research indicates that elevated PARP activity during chronic DNA damage and rising CD38 expression with age both contribute to NAD+ depletion in tissues.
The NAD+ salvage pathway regenerates NAD+ from nicotinamide via NAMPT (nicotinamide phosphoribosyltransferase) as the rate-limiting enzyme. Research into NAD+ precursors like NMN and NR focuses on feeding this salvage pathway indirectly. Direct NAD+ is studied at the molecular and cellular level for its role in restoring coenzyme pools in models where salvage capacity is limited.
Peptideware supplies NAD+ 500mg as a lyophilized research powder, reconstituted with bacteriostatic water for laboratory protocols investigating bioenergetic decline, sirtuin substrate availability, and mitochondrial biogenesis markers in preclinical systems.
How Does SS-31 (Elamipretide) Function?
SS-31 belongs to the Szeto-Schiller peptide class, a family of aromatic-cationic tetrapeptides engineered by Hazel Szeto and Peter Schiller to cross cell membranes and localize to the inner mitochondrial membrane (IMM). Its sequence — D-Arg-2′,6′-Dmt-Lys-Phe-NH2 — includes a dimethyltyrosine residue and D-amino acids that resist enzymatic degradation.
The defining mechanistic feature of SS-31 is its selective affinity for cardiolipin. Published studies suggest SS-31 concentrates more than 1,000-fold in the IMM through electrostatic and hydrophobic interactions with cardiolipin’s unique four-acyl-chain structure. By associating with cardiolipin, SS-31 appears to protect it from peroxidation, preserve cristae curvature, and stabilize ETC supercomplexes (I-III-IV) that depend on intact cardiolipin scaffolding.
Preclinical research indicates SS-31 supports mitochondrial membrane potential and reduces ROS leakage in models of ischemia-reperfusion, heart failure, and age-related skeletal muscle decline. Unlike antioxidants that scavenge ROS after generation, SS-31 is hypothesized to reduce ROS generation upstream by maintaining the structural integrity of the respiratory chain.
Peptideware supplies SS-31 10mg as a lyophilized peptide for research use. The 10mg format reflects the peptide’s potency in preclinical models, where effective concentrations are typically studied in the low micromolar range.
What Does Published Research Show?
Published studies on NAD+ biology span multiple decades. Work from the laboratories of Leonard Guarente, David Sinclair, Shin-ichiro Imai, and Johan Auwerx established that sirtuin activation depends on NAD+ availability, and that raising cellular NAD+ in mouse models supports mitochondrial biogenesis, improves insulin sensitivity, and extends healthspan metrics. PubMed-indexed reviews (e.g., Verdin 2015, Science; Covarrubias et al. 2021, Nature Reviews Molecular Cell Biology) describe the NAD+ decline observed in aged tissues and the molecular consequences of NAMPT downregulation.
Preclinical research on NAD+ precursors has moved into early human research contexts for conditions like mitochondrial myopathy, though direct NAD+ research remains largely at the cellular and animal model stage due to bioavailability challenges.
SS-31 research has progressed differently. Foundational work by Szeto and colleagues (Zhao et al. 2004, Journal of Biological Chemistry; Szeto 2014, British Journal of Pharmacology) characterized the cardiolipin-binding mechanism. Preclinical studies in ischemia-reperfusion models demonstrated preservation of mitochondrial respiration and reduced infarct size. Under the development name Elamipretide, the compound has been evaluated in research programs targeting Barth syndrome, primary mitochondrial myopathy, and dry age-related macular degeneration in controlled studies.
Both research streams converge on a shared conclusion: mitochondrial dysfunction is a modifiable variable in preclinical models of aging and disease, and distinct molecular strategies (coenzyme replenishment vs. structural stabilization) produce distinct downstream signatures in tissues studied.
Which Approach for Which Research?
Choosing between NAD+ and SS-31 in a research protocol depends on the biological question being asked.
NAD+ is typically studied when:
- The research question centers on the sirtuin pathway or SIRT1/SIRT3 activity in tissues
- Models involve NAD+ depletion from chronic PARP activation or CD38 overexpression
- Readouts include mitochondrial biogenesis markers (PGC-1α, TFAM, NRF1)
- The protocol examines bioenergetic substrate availability at the whole-cell level
- Research explores aging-associated metabolic dysfunction in preclinical models
SS-31 is typically studied when:
- The focus is on cardiolipin integrity or cristae ultrastructure
- Models involve ischemia-reperfusion, acute oxidative stress, or cardiac preclinical contexts
- Readouts include mitochondrial membrane potential (ΔΨm), ETC supercomplex assembly, or ROS production rates
- Research targets conditions with documented cardiolipin peroxidation
- A targeted peptide delivery approach is preferred over broad coenzyme replenishment
Many research programs explore both molecules in parallel or sequential protocols to distinguish structural mitochondrial effects (SS-31) from bioenergetic and transcriptional effects (NAD+). The two approaches are not mutually exclusive, and preclinical research indicates they operate on complementary aspects of mitochondrial biology. A decision matrix can help research teams scope protocols: if the primary endpoint is NAD+/NADH ratio, sirtuin deacetylation activity, or transcriptional output from PGC-1α and related mitochondrial biogenesis factors, NAD+ is the more direct tool. If the primary endpoint is cristae morphology by electron microscopy, cardiolipin peroxidation by mass spectrometry, or supercomplex assembly by blue native gel electrophoresis, SS-31 is the more direct tool. Protocols targeting overall mitochondrial respiration via Seahorse analysis may incorporate either or both compounds depending on the hypothesized upstream mechanism.
Frequently Asked Research Questions
Is NAD+ actually a peptide?
No. NAD+ (nicotinamide adenine dinucleotide) is a dinucleotide coenzyme, not a peptide. It consists of two nucleotides — adenine mononucleotide and nicotinamide mononucleotide — joined through their phosphate groups. Peptides, by contrast, are short chains of amino acids linked by peptide bonds. We include NAD+ in peptide-focused research comparisons because laboratory workflows for reconstitution, storage, and research handling overlap with those used for peptide research compounds, and because NAD+ is frequently studied alongside peptides in mitochondrial research programs. From a strict chemistry standpoint, however, NAD+ belongs to the nucleotide cofactor class. Any literature or supplier describing NAD+ as a “peptide” is using the term loosely, and researchers should be aware of this distinction when categorizing compounds in protocols and documentation.
Why target cardiolipin specifically?
Cardiolipin is a phospholipid unique to the inner mitochondrial membrane, and it serves as a structural scaffold for electron transport chain supercomplexes. Published studies suggest cardiolipin molecules cluster around Complexes I, III, and IV, and their presence is required for optimal supercomplex assembly and function. Cardiolipin is also highly susceptible to peroxidation because its four acyl chains contain polyunsaturated fatty acids positioned close to the primary site of ROS generation. When cardiolipin is peroxidized, cristae architecture collapses, cytochrome c can be released, and respiratory efficiency drops. Preclinical research indicates that compounds which bind cardiolipin without disrupting its function can preserve cristae morphology and maintain respiratory chain integrity — which is why SS-31’s cardiolipin affinity is considered mechanistically significant in mitochondrial research contexts.
How do these molecules get into mitochondria?
The two compounds use entirely different entry routes. NAD+ cellular uptake is limited because the intact dinucleotide does not readily cross plasma membranes; most research explores precursors (NMN, NR) that cross membranes and are converted to NAD+ intracellularly, or examines NAD+ effects in permeabilized cell systems and isolated mitochondria. SS-31, by contrast, was engineered with aromatic-cationic properties that enable it to cross plasma and mitochondrial membranes in a potential-independent manner. Published studies suggest SS-31 accumulates in the inner mitochondrial membrane more than 1,000-fold relative to cytosolic concentrations. This fundamental difference — indirect cofactor replenishment versus direct IMM targeting — is one reason the two compounds are often studied using different protocol designs in preclinical research.
Can both be studied in the same research protocol?
Yes. Preclinical research programs sometimes combine NAD+ (or NAD+ precursors) with SS-31 or related Szeto-Schiller peptides to investigate additive or synergistic effects on mitochondrial endpoints. The rationale is mechanistic complementarity: NAD+ addresses coenzyme availability for ETC flux and sirtuin signaling, while SS-31 addresses structural membrane integrity. When designing combination protocols, researchers typically account for distinct reconstitution requirements, stability profiles, and administration schedules of each compound. Appropriate controls — including single-compound arms — are essential to attribute observed effects correctly. Published studies on combination approaches remain less common than single-compound research, so laboratories conducting parallel protocols often contribute novel data to the field. All such work is strictly for laboratory and research purposes.
What about stability and reconstitution?
NAD+ is relatively hygroscopic and sensitive to heat and prolonged light exposure. Lyophilized NAD+ powder is typically stored at -20°C long-term and reconstituted with bacteriostatic water shortly before research use. Reconstituted NAD+ solutions have limited stability and are often prepared fresh for each experimental session. SS-31 lyophilized peptide is also stored at -20°C; reconstituted SS-31 in bacteriostatic water is generally more stable than NAD+ in solution but still benefits from refrigerated storage (2-8°C) and protection from repeated freeze-thaw cycles. Specific stability data for each batch should be referenced from certificates of analysis. Both compounds are supplied strictly for laboratory and research purposes and should be handled using appropriate research-grade protocols and personal protective equipment.
Which has more published human research?
SS-31 (under the development name Elamipretide) has moved further along the clinical research continuum for specific indications. Research programs have examined Elamipretide in studies involving Barth syndrome, primary mitochondrial myopathy, and dry age-related macular degeneration, generating peer-reviewed publications describing outcomes in controlled research settings. NAD+ human research is more fragmented: most published human work focuses on NAD+ precursors (NMN, NR) rather than direct NAD+, because of bioavailability considerations. At the cellular and animal model level, NAD+ has a much larger body of preclinical literature spanning sirtuin biology, metabolism, and aging research. Neither compound is approved for general human use, and products supplied by Peptideware are intended exclusively for laboratory and research purposes. Researchers should consult PubMed directly for the most current publication landscape.
Related Longevity Research Reading
For a broader overview of longevity-focused research compounds, see the Longevity Peptides Research Guide, which covers NAD+ biology, Szeto-Schiller peptides, senolytic approaches, and related research categories in greater depth.
All products are intended for laboratory and research purposes only. Not for human or animal consumption. These statements have not been evaluated by the FDA.