Last Updated: April 14, 2026
Incretin-based research peptides have reshaped preclinical metabolic science over the past decade. Tirzepatide and retatrutide are two of the most studied next-generation incretin mimetics, each engineered to engage multiple metabolic receptors simultaneously. Tirzepatide operates as a dual agonist targeting the glucose-dependent insulinotropic polypeptide (GIP) receptor and the glucagon-like peptide-1 (GLP-1) receptor, while retatrutide extends that mechanism by adding glucagon receptor (GCGR) activity, making it a triple agonist. Researchers comparing these molecules in laboratory use are generally examining how receptor plurality affects glycemic parameters, body composition endpoints, energy expenditure signaling, and pharmacokinetics in preclinical models. This article summarizes the receptor pharmacology, structural properties, and published study landscape for tirzepatide vs retatrutide, and outlines considerations for researchers selecting between them for protocol design. All information below is compiled for laboratory and research purposes only and references published PubMed-indexed literature where applicable.
Quick Comparison Table
| Attribute | Tirzepatide | Retatrutide |
|---|---|---|
| Receptor targets | GIP + GLP-1 (dual) | GLP-1 + GIP + GCGR (triple) |
| Molecular weight | ~4,813 Da | ~4,731 Da |
| Amino acid count | 39 residues | 39 residues |
| Reported half-life (preclinical) | ~5 days | ~6 days |
| Fatty acid modification | C20 diacid (albumin binding) | C20 diacid (albumin binding) |
| Typical research dosing range (in vivo studies) | 2.5–15 mg weekly equivalents reported | 1–12 mg weekly equivalents reported |
| Development stage (as of 2026) | Approved agent; extensively published | Phase 3 programs reported; Phase 2 data published |
What Is a Dual Incretin Agonist?
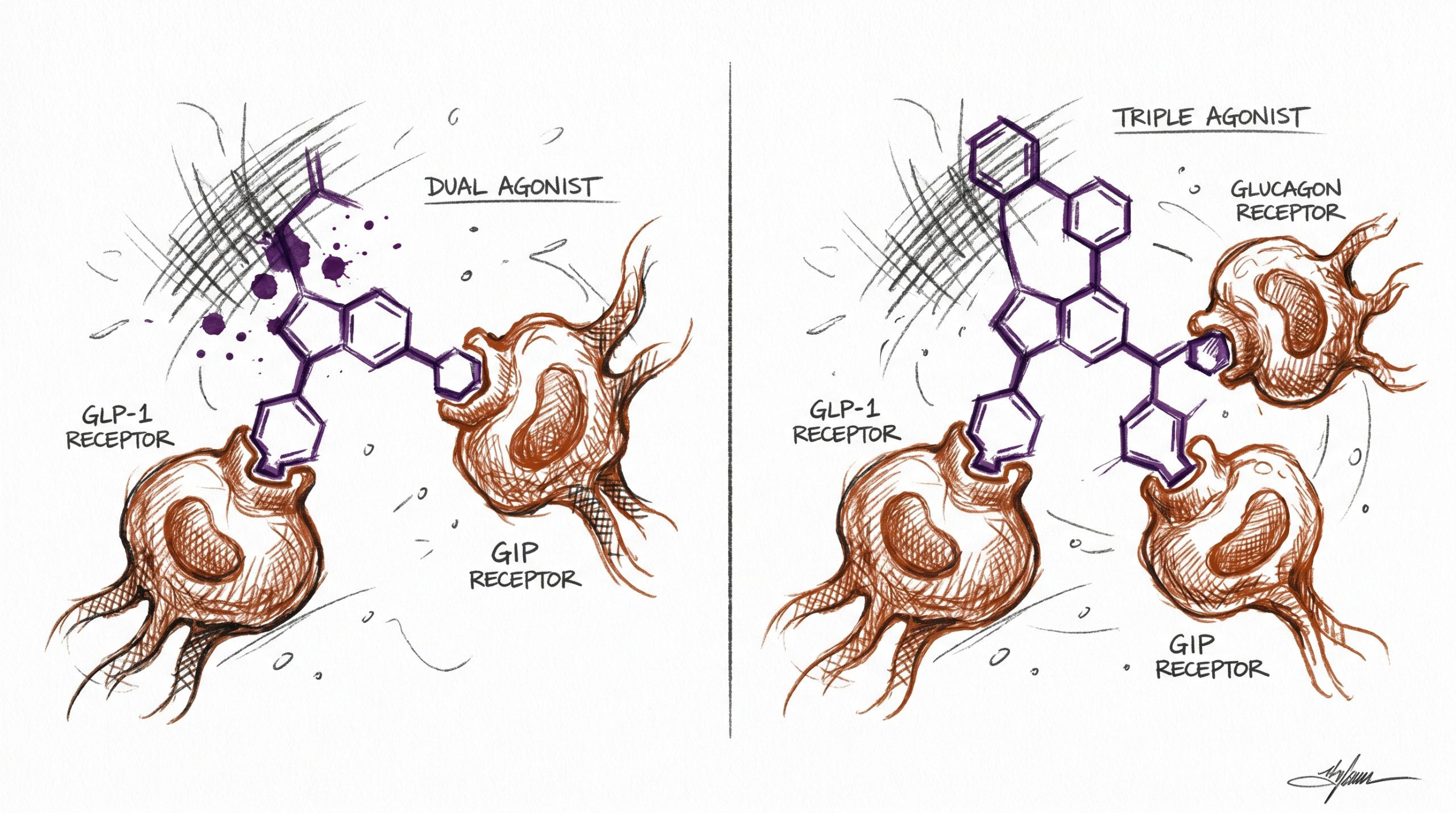
A dual incretin agonist is a single peptide designed to activate two gut-hormone receptors at once. Tirzepatide, the best-characterized example, engages both the GIP and GLP-1 receptors through a unified 39-amino-acid backbone with a C20 fatty diacid conjugated to a lysine residue, which extends its circulating half-life by binding albumin. The dual-receptor concept, often called the “twincretin” approach, was built on preclinical observations that GIP and GLP-1 contribute complementary insulinotropic and satiety-related signaling in rodent models.
In published research, tirzepatide has demonstrated superior glycemic and body-weight effects compared with selective GLP-1 agonists in head-to-head studies. The SURPASS-2 trial (PubMed ID 34170647) reported greater HbA1c reductions with tirzepatide versus semaglutide 1 mg in type 2 diabetes cohorts, while SURMOUNT-1 (PubMed ID 35658024) documented substantial body-weight reductions versus placebo in obesity cohorts. Published studies suggest GIP receptor agonism may amplify GLP-1-driven effects on adipose tissue handling in preclinical research, though the exact contribution of GIP signaling remains an active area of investigation. For researchers, tirzepatide represents a well-characterized reference compound for dual-incretin comparative pharmacology.
What Is a Triple Agonist?
A triple agonist extends the incretin concept by adding a third receptor target. Retatrutide (also known by its development code LY3437943) engages GLP-1, GIP, and the glucagon receptor (GCGR) through a single 39-amino-acid sequence that shares structural homology with tirzepatide but features amino acid substitutions that restore glucagon-receptor affinity. The rationale for adding GCGR activity is rooted in preclinical research indicating that glucagon-receptor signaling increases resting energy expenditure and hepatic lipid mobilization in rodent models, effects that are not produced by pure incretin agonism.
The theoretical appeal of triple agonism is that GLP-1 drives satiety and glycemic control, GIP enhances insulin response and may modulate adipose function, and GCGR activity raises energy expenditure. Preclinical research indicates that balanced tri-agonism can produce greater reductions in adiposity than dual agonism at matched exposures in animal obesity models. Published Phase 2 data for retatrutide in obesity (PubMed ID 37351564) and type 2 diabetes (PubMed ID 37385275) reported dose-dependent reductions in body weight and glycated hemoglobin across 48 weeks. The challenge in triple-agonist design is balancing GCGR activity, because excessive glucagon signaling can offset incretin-mediated glycemic benefits; retatrutide’s published profile reflects careful tuning of this balance.
How Do These Compounds Compare in Published Research?
The published literature comparing tirzepatide and retatrutide is anchored by four pivotal studies, summarized below. Researchers designing comparative protocols typically reference these datasets to frame expected magnitudes of effect and to structure dose-response arms.
| Study | Compound | Population/Model | Duration | PubMed |
|---|---|---|---|---|
| SURMOUNT-1 | Tirzepatide | Adults with obesity (n=2,539) | 72 weeks | 35658024 |
| SURPASS-2 | Tirzepatide vs semaglutide | Adults with type 2 diabetes (n=1,879) | 40 weeks | 34170647 |
| Retatrutide Phase 2 (Obesity) | Retatrutide | Adults with obesity (n=338) | 48 weeks | 37351564 |
| Retatrutide Phase 2 (T2D) | Retatrutide | Adults with type 2 diabetes (n=281) | 36 weeks | 37385275 |
Published studies suggest retatrutide produced larger mean body-weight reductions at top reported doses than tirzepatide in cohorts with obesity, although direct head-to-head trials have not been published at time of writing. Both compounds showed dose-dependent glycemic improvements in type 2 diabetes cohorts. For researchers, the implication is that retatrutide’s additional GCGR arm appears to contribute incremental effects beyond dual incretin agonism alone, consistent with preclinical energy-expenditure hypotheses, though cross-study comparisons must be interpreted with caution because trial designs, cohorts, and endpoints differ.
Researchers comparing the pharmacokinetic behavior of tirzepatide and retatrutide in preclinical models should note that both compounds exhibit slow absorption profiles following subcutaneous administration, with reported time-to-peak concentrations in the 24 to 72 hour range and steady-state reached after several weekly-equivalent administrations. The C20 diacid moiety on both peptides is the primary determinant of extended half-life because albumin binding reduces renal filtration and proteolytic clearance. Preclinical research indicates that receptor-specific activity at each of the targeted receptors can be characterized using standard cAMP accumulation assays in transfected cell lines, and that retatrutide’s relative receptor potency profile is typically reported as balanced across GLP-1, GIP, and GCGR, whereas tirzepatide shows strong GIP and GLP-1 activity with minimal GCGR engagement. For comparative in vivo pharmacology, matching exposures rather than nominal doses is generally considered the more rigorous approach because peptide bioavailability and clearance can differ across animal models.
Which Should Researchers Choose?
The selection between tirzepatide and retatrutide depends on the research objective. The framework below maps common preclinical aims to the more appropriate reference compound.
- Comparative pharmacokinetic profiling: Both peptides share a 39-residue backbone and C20 fatty acid linker, making them useful paired references for albumin-binding and half-life studies. Tirzepatide is the better-characterized starting point because more public PK data exist.
- Incretin-only mechanism studies: If the objective is to isolate GIP + GLP-1 effects without glucagon-receptor confounding, tirzepatide is the cleaner reference compound. Retatrutide’s GCGR activity would introduce an additional variable.
- Energy expenditure and metabolic rate endpoints: Retatrutide is the more informative compound because GCGR signaling is mechanistically linked to thermogenesis and hepatic substrate handling in preclinical models.
- Adiposity and body-composition endpoints: Both are relevant; published studies suggest retatrutide produces larger mean effects at top doses, while tirzepatide offers deeper published context for longitudinal comparisons.
- Receptor selectivity or crosstalk studies: Tirzepatide is a better tool for dissecting GIP/GLP-1 crosstalk; retatrutide is essential if GCGR crosstalk is the focus.
For laboratories designing comparative incretin research protocols, running both compounds as parallel reference arms is increasingly common because it isolates the contribution of GCGR activity while keeping the incretin scaffold matched. A well-structured comparative protocol typically holds dose-matching, administration schedule, animal model selection, and endpoint cadence constant across arms, leaving receptor pharmacology as the primary independent variable. This design approach, grounded in published study precedent, allows the incremental contribution of glucagon-receptor activity to be quantified against a well-characterized dual-agonist baseline. Both compounds are available for laboratory use at Tirzepatide and Retatrutide, and broader incretin context is covered on the GLP-1 agonists comparison pillar page alongside semaglutide research context.
Frequently Asked Questions
What’s the difference between dual and triple agonism?
Dual agonism refers to a single peptide engaging two receptors at once; triple agonism extends that to three. In the tirzepatide vs retatrutide comparison, tirzepatide activates the GIP and GLP-1 receptors, while retatrutide adds the glucagon receptor (GCGR). The practical implication for research protocols is that retatrutide introduces an additional signaling pathway linked to energy expenditure and hepatic substrate mobilization in preclinical models. Dual agonism is useful when the research objective is to characterize incretin-only mechanisms, whereas triple agonism is more appropriate when the research design specifically aims to probe glucagon-receptor contribution to metabolic endpoints. Neither is inherently superior; the optimal selection depends on the hypothesis being tested and the endpoints under measurement. Published studies suggest the magnitude of effect in obesity cohorts may be larger with triple agonism at top doses, though head-to-head laboratory comparisons are still emerging in the published record.
Why does GCGR addition matter?
The glucagon receptor, historically viewed as a counter-regulatory target opposing insulin, has a second role as a driver of resting energy expenditure and hepatic lipid handling. Preclinical research indicates that controlled GCGR activation increases mitochondrial activity, fatty acid oxidation, and hepatic glycogenolysis in rodent models. When paired with GLP-1-mediated insulin secretion, the glycemic destabilization typically associated with pure glucagon agonism is offset, allowing the energy-expenditure benefits to manifest without the counter-regulatory downside. This is the design rationale behind retatrutide’s tri-agonist structure. For researchers, the inclusion of GCGR provides an experimental handle on the thermogenic axis that incretin-only compounds cannot engage. Published Phase 2 data (PubMed ID 37351564) suggest this translates to greater body-weight reductions at matched durations in obesity cohorts. In comparative protocols, GCGR addition is the specific variable under test when retatrutide is placed alongside tirzepatide.
How are these peptides stored for research?
Both tirzepatide and retatrutide are lyophilized powders that should be stored at -20 degrees Celsius prior to reconstitution for research. Once reconstituted in bacteriostatic water or sterile saline for laboratory use, they are typically stored at 2 to 8 degrees Celsius and used within the stability window reported on the certificate of analysis. Repeat freeze-thaw cycles should be avoided because peptide bond degradation and aggregation can compromise assay results. Researchers should protect reconstituted solutions from light and handle them under aseptic technique to maintain integrity. For long-term storage of stock material, single-use aliquots at -80 degrees Celsius are preferred in many laboratories. Always refer to the compound-specific certificate of analysis and any supplier documentation for detailed storage and handling guidance, and document reconstitution date and concentration in laboratory records to maintain experimental reproducibility across comparative arms.
Are tirzepatide and retatrutide structurally similar?
Yes, both are 39-amino-acid peptides with a C20 fatty diacid conjugated to a lysine residue that enables albumin binding and extends circulating half-life. Their primary sequences share substantial homology because both were engineered from the broader exendin/GIP scaffold tradition. The critical structural difference lies in specific residue substitutions that modulate receptor affinity. Tirzepatide is optimized for GIP and GLP-1 receptor engagement with minimal glucagon-receptor activity, while retatrutide retains GLP-1 and GIP affinity but introduces substitutions that restore meaningful glucagon-receptor binding. Molecular weights are similar, with tirzepatide around 4,813 Daltons and retatrutide around 4,731 Daltons. This structural parallelism makes them excellent companion reference compounds in preclinical research because many physicochemical properties such as solubility, albumin binding, and subcutaneous depot behavior are comparable, isolating receptor pharmacology as the primary experimental variable.
What endpoints are typically measured in preclinical research with these compounds?
Common endpoints in preclinical research with tirzepatide and retatrutide include body weight trajectories, body composition via DEXA or MRI in animal models, fasting and postprandial glucose, HbA1c or equivalent glycemic markers, plasma insulin and glucagon dynamics, hepatic triglyceride content, and indirect calorimetry measures such as resting energy expenditure and respiratory exchange ratio. Researchers interested in the GCGR-specific contribution often add hepatic gene expression panels covering gluconeogenic and lipid-oxidation pathways. Pharmacokinetic endpoints include area under the curve, peak concentration, half-life, and albumin-binding fraction. Behavioral endpoints such as food intake patterns and satiety-proxy measures are also common. Published studies suggest both compounds produce dose-dependent effects across these domains in preclinical models, with retatrutide showing additional signal on energy-expenditure readouts consistent with its GCGR arm. Endpoint selection should align with the specific receptor mechanism being interrogated in the protocol.
Which compound has more published research data available?
Tirzepatide has substantially more published research data as of 2026 because it entered clinical development earlier and has a larger completed trial program across the SURPASS and SURMOUNT study families covering type 2 diabetes, obesity, sleep-disordered breathing, and cardiometabolic endpoints. The peer-reviewed record for tirzepatide includes multiple Phase 3 trials, real-world observational cohorts, and mechanistic pharmacology studies. Retatrutide’s published record is growing rapidly, anchored by Phase 2 obesity and type 2 diabetes results (PubMed IDs 37351564 and 37385275), with additional Phase 3 datasets expected as trials report out. For researchers starting a comparative protocol, the practical implication is that tirzepatide offers a broader reference base for benchmarking assay performance and expected magnitudes of effect, while retatrutide provides a more mechanistically novel reference for triple-agonist work. Both compounds have well-characterized certificates of analysis available for research-grade material.
All products are intended for laboratory and research purposes only. Not for human or animal consumption. These statements have not been evaluated by the FDA.