Last Updated: April 14, 2026
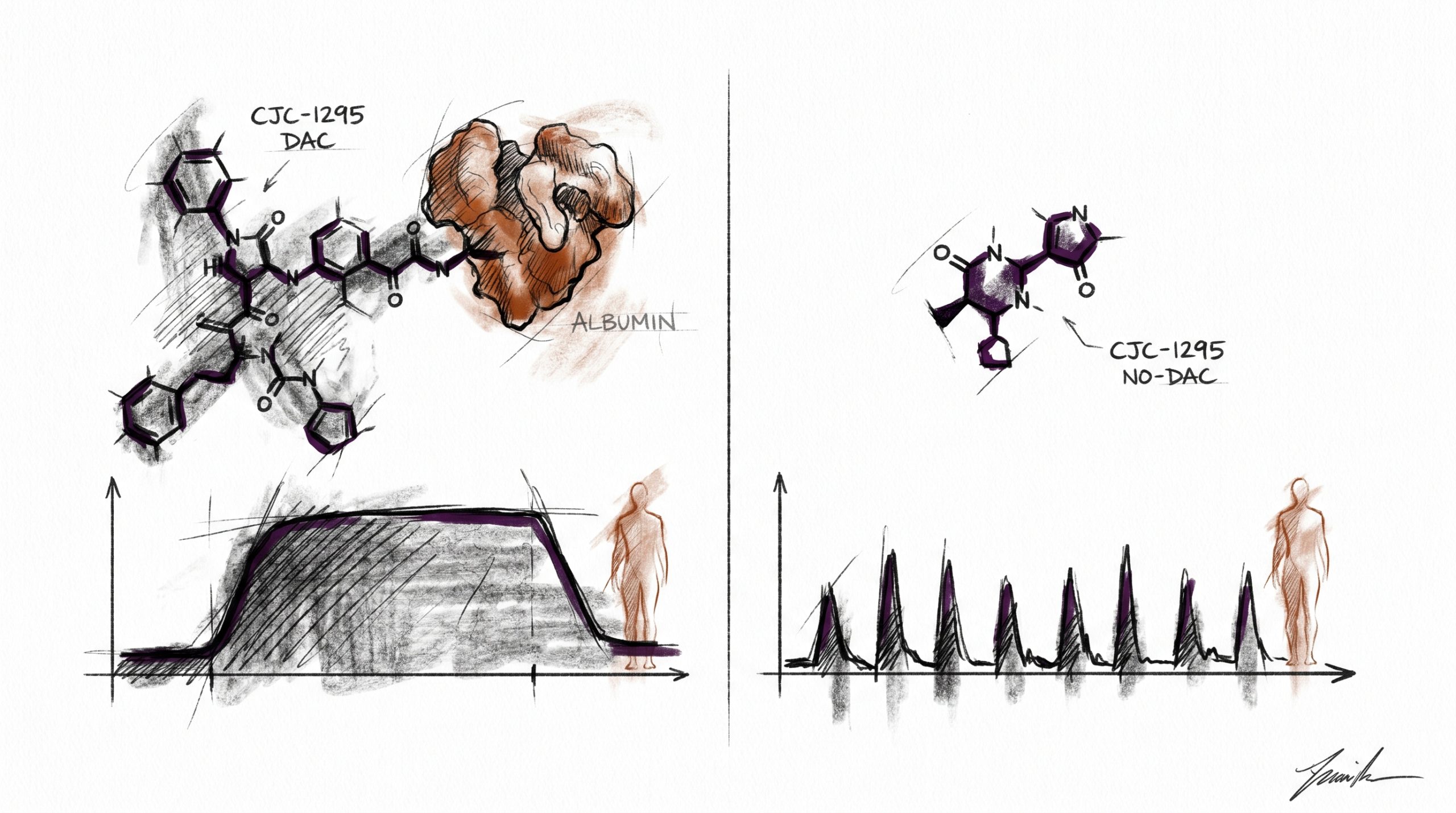
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH) that has become a standard reference compound in endocrine research examining the somatotropic axis. The peptide exists in two distinct research forms that share the same 30-amino-acid core sequence but differ in a single pharmacokinetic modification: one version carries a Drug Affinity Complex (DAC) linker, and the other does not. That one structural difference changes circulating half-life from roughly 30 minutes to approximately 8 days, and it changes the resulting growth hormone (GH) release profile from a pulsatile pattern resembling endogenous secretion to a sustained elevation of GH and IGF-1. This comparison is written for laboratory researchers evaluating which variant fits a given in vitro assay or preclinical model. It does not constitute guidance for human or animal administration. All products referenced are intended for laboratory and research purposes only.
Quick Comparison: CJC-1295 DAC vs No DAC
| Attribute | CJC-1295 With DAC | CJC-1295 No DAC (Mod GRF 1-29) |
|---|---|---|
| Half-life | ~8 days (approximately 6-8 days in preclinical PK studies) | ~30 minutes |
| GH release pattern | Sustained elevation of GH and IGF-1 | Pulsatile, short-duration GH release |
| Albumin binding | Covalent, via maleimidopropionic acid (MPA) linker | None |
| Structural modification | DAC/MPA linker at C-terminus; 4 amino acid substitutions for stability | 4 amino acid substitutions only (no linker) |
| Research application | Sustained-exposure pharmacokinetic models; IGF-1 bioavailability studies | Physiological pulsatile-mimicry protocols; short-window receptor studies |
| Typical co-investigation | Longitudinal endocrine assays | Synergy research with GH secretagogues (e.g., Ipamorelin) |
What Is a GHRH Analog?
Growth hormone-releasing hormone is a 44-amino-acid hypothalamic peptide that binds the GHRH receptor (GHRH-R) on anterior pituitary somatotrophs. Binding activates a Gs-coupled signaling cascade, elevates intracellular cAMP, and triggers release of stored growth hormone into portal circulation. Endogenous GHRH is released in pulses synchronized with somatostatin troughs, producing the episodic GH secretion pattern observed in preclinical animal models and characterized in published endocrinology literature.
GHRH analogs are engineered peptides that preserve the receptor-binding pharmacophore — typically the N-terminal 29 amino acids, which constitute the minimum sequence for full biological activity — while substituting or modifying residues to resist enzymatic degradation. Native GHRH is cleaved rapidly by dipeptidyl peptidase-4 (DPP-4) at the Ala2-Asp3 bond, giving it a plasma half-life under 7 minutes. Analogs substitute D-Ala at position 2, Gln at position 8, Ala at position 15, and Leu at position 27 to block proteolysis and reduce methionine oxidation. The tetrasubstituted sequence is commonly designated “Modified GRF 1-29” or “CJC-1295 without DAC.” Adding the MPA/DAC linker to that same sequence produces “CJC-1295 with DAC.” Both variants are used in laboratory research to probe somatotroph function under controlled conditions.
How Does CJC-1295 With DAC Work?
The defining feature of CJC-1295 with DAC is the Drug Affinity Complex — a maleimidopropionic acid (MPA) moiety conjugated to the lysine residue at position 30 of the modified GRF 1-29 sequence. The MPA group contains a maleimide functional group that reacts covalently with the free thiol of cysteine-34 on circulating serum albumin. Because albumin has a plasma half-life of approximately 19 days in preclinical mammalian models, tethering the peptide to albumin dramatically extends its circulating lifetime.
In published pharmacokinetic research, the DAC-modified peptide exhibits a terminal half-life in the range of 6-8 days. This produces a bioactive GHRH signal that persists for days rather than minutes. Preclinical research indicates that sustained GHRH-R stimulation elevates mean GH concentrations and raises IGF-1 approximately two- to three-fold above baseline for extended periods. The pattern is not pulsatile — it approximates a continuous infusion, which makes this variant useful for research questions involving long-duration somatotroph stimulation, IGF-1 downstream signaling studies, and pharmacokinetic modeling of depot-like peptide behavior.
The trade-off is that continuous stimulation departs from the natural secretion pattern. For research protocols where physiological mimicry matters, this is a limitation; for protocols that isolate the effect of prolonged GHRH-R activation, it is the entire point.
How Does CJC-1295 Without DAC (Mod GRF 1-29) Work?
CJC-1295 without DAC — also catalogued as Modified GRF 1-29 — is the same 29-amino-acid sequence with the same four stabilizing substitutions, but without the MPA linker. Because there is no albumin tether, the peptide circulates freely and is cleared through renal filtration and residual proteolysis within approximately 30 minutes of administration in preclinical models.
That short exposure window produces a discrete pulse of somatotroph activation. GH rises sharply, reaches peak concentration within roughly 30 minutes, and returns toward baseline within 2-3 hours. The resulting GH envelope more closely resembles endogenous pulsatile secretion than the sustained elevation produced by the DAC variant. In preclinical research examining pulse frequency, amplitude modulation, or the interaction between GHRH signaling and somatostatin feedback, the no-DAC variant is generally preferred because it preserves the temporal structure of the signal.
This variant is also the default choice in research combining a GHRH analog with a growth hormone secretagogue receptor (GHS-R) agonist, because the short GHRH pulse can be co-timed with the ghrelin-mimetic pulse to study synergistic release patterns without the confound of a persistent GHRH background.
When Is Each Variant Used in Research?
The selection framework is driven by what the research question requires. Published studies suggest the following decision criteria:
- Choose CJC-1295 with DAC when: the protocol calls for multi-day sustained GHRH-R activation, when IGF-1 steady-state elevation is the measured outcome, when infrequent administration in a preclinical model is a practical constraint, or when the research models depot pharmacokinetics.
- Choose CJC-1295 without DAC when: the protocol depends on pulsatile delivery, when precise timing of GH release relative to another stimulus is needed, when measuring acute somatotroph responsiveness rather than chronic adaptation, or when combining the GHRH analog with a GHS-R agonist.
The most common combination protocol pairs a short-acting GHRH analog with a selective ghrelin receptor agonist. Ipamorelin is frequently used in this role because it activates GHS-R1a without measurable effects on cortisol, prolactin, or ACTH in preclinical research — making it a clean probe for isolating the GH axis. Pairing Ipamorelin with CJC-1295 no DAC produces a coordinated dual-pulse on the two complementary receptors and is a standard comparator arm in somatotroph synergy research. Pairing Ipamorelin with CJC-1295 DAC is less common because the sustained DAC background obscures the pulse structure that the combination is typically designed to study.
What Does Published Research Show?
The original pharmacokinetic characterization of the DAC modification was published by Teichman and colleagues (PubMed: 16352685), which documented the extended half-life of the albumin-tethered peptide and demonstrated dose-dependent elevations of GH and IGF-1 across the dosing window in a preclinical pharmacokinetic study. Subsequent receptor-binding work (Jetté et al., PubMed: 15845616) confirmed that GHRH-R selectivity is preserved after MPA conjugation and that the peptide retains nanomolar affinity for the receptor.
For the unmodified tetrasubstituted sequence, the foundational pharmacology of the substitutions is rooted in earlier GHRH analog research going back to the 1990s, which established that D-Ala2, Gln8, Ala15, and Leu27 confer resistance to DPP-4 cleavage and oxidation without compromising receptor activation (representative work includes Coy et al. and Robberecht et al., indexed in PubMed).
Contemporary preclinical research continues to use both variants as pharmacological tools: the DAC form as a long-acting GHRH-R probe, and the no-DAC form as a short-acting tool compatible with pulsatile assay designs. Neither variant is FDA-approved for human use. Both are classified as research chemicals intended for laboratory use only.
Frequently Asked Questions
What does DAC stand for?
DAC stands for Drug Affinity Complex. In the CJC-1295 context, it refers to a maleimidopropionic acid (MPA) linker conjugated to the C-terminal lysine of the modified GRF 1-29 peptide sequence. The maleimide functional group of the MPA linker reacts covalently with the free thiol on cysteine-34 of circulating serum albumin, forming a stable thioether bond. Because albumin has a plasma half-life of approximately 19 days in preclinical mammalian models, tethering the peptide to albumin extends the peptide’s effective circulating lifetime from roughly 30 minutes to approximately 6-8 days. The DAC technology is a platform approach to peptide half-life extension and has been applied to other peptides beyond CJC-1295 in published pharmaceutical research literature. All DAC-modified research peptides remain classified as laboratory compounds and are intended for laboratory use only.
Why would a researcher choose the shorter half-life variant?
Researchers select the no-DAC variant when the experimental design requires pulsatile signaling that mimics endogenous GHRH release, when precise temporal control matters, or when combining a GHRH analog with a GHS-R agonist for synergy research. The short half-life means each administration produces a discrete GH pulse followed by a return to baseline, preserving the oscillatory structure of the somatotropic axis. This is essential in preclinical research examining pulse frequency, amplitude modulation, somatostatin feedback dynamics, and receptor desensitization kinetics. Sustained GHRH-R activation from the DAC variant would overwrite the natural pulse pattern and confound these measurements. The shorter half-life also allows researchers to observe acute somatotroph responsiveness without the carryover effects that persist for days with the DAC form. Research protocols studying physiological-mimicry endocrine models almost always use the no-DAC variant for these reasons.
Can these be combined with Ipamorelin?
In published preclinical research and laboratory protocols, Ipamorelin is most commonly paired with CJC-1295 without DAC rather than with the DAC variant. The reason is mechanistic: Ipamorelin is a selective agonist of the growth hormone secretagogue receptor (GHS-R1a), which is a different receptor from GHRH-R, and the two receptors activate complementary intracellular pathways in somatotrophs. Co-stimulation of both receptors produces greater GH release than either alone in preclinical models. For this synergy to be measurable as a discrete pulse, both signals need to be time-aligned and transient — which requires a short-acting GHRH analog. The DAC variant produces a sustained GHRH-R background that masks the pulse structure and is therefore less suited to combination research. Preclinical research indicates that the Ipamorelin plus no-DAC CJC-1295 pairing is the standard laboratory comparator for dual-receptor GH release studies.
Are the two variants structurally different beyond the linker?
No. The core 29-amino-acid sequence and the four stabilizing amino acid substitutions — D-Ala at position 2, Gln at position 8, Ala at position 15, and Leu at position 27 — are identical in both variants. The only structural difference is the presence or absence of the maleimidopropionic acid (MPA) linker conjugated to a C-terminal lysine extension. The DAC variant has this linker; the no-DAC variant does not. Because the receptor-binding pharmacophore is unchanged, both variants activate the GHRH receptor with comparable nanomolar affinity in published binding assays. What differs is pharmacokinetics: the DAC variant binds circulating albumin covalently and persists for days, while the no-DAC variant is cleared within approximately 30 minutes. Researchers should treat the two compounds as pharmacokinetic variants of a single pharmacological agent rather than as distinct peptides.
How is IGF-1 response different between the variants?
Published research indicates that CJC-1295 with DAC produces a sustained elevation of IGF-1 — typically two- to three-fold above baseline — maintained over several days in preclinical pharmacokinetic models, because continuous GHRH-R stimulation drives continuous GH release, and GH in turn drives hepatic IGF-1 production with a lag of roughly 24 hours. The no-DAC variant produces only transient GH pulses, and although each pulse contributes to IGF-1 production, the short exposure window produces smaller and less sustained IGF-1 changes. Research protocols that measure IGF-1 as the primary endpoint generally use the DAC variant because the sustained elevation is easier to quantify. Protocols that measure acute GH release as the primary endpoint generally use the no-DAC variant because each administration produces a clean pulse. Preclinical research indicates that the choice depends on whether the research question is about acute signaling or chronic exposure.
Are CJC-1295 variants approved for any clinical use?
No. Neither CJC-1295 with DAC nor CJC-1295 without DAC is approved by the FDA or comparable regulatory bodies for any clinical indication. Both compounds are classified as research chemicals intended for laboratory use only. Published research on these peptides is limited to preclinical pharmacokinetic and pharmacodynamic studies and early-phase exploratory work; no large-scale clinical programs have progressed to approval. Products sold as CJC-1295 DAC or CJC-1295 no DAC through research chemical suppliers are not pharmaceutical products and are not intended for human or animal consumption. Laboratories working with these compounds should follow standard research chemical handling protocols, maintain documented chain of custody, and reconstitute for research using bacteriostatic water or other appropriate solvents per their institutional guidelines. These statements have not been evaluated by the FDA, and none of the information in this article should be interpreted as guidance for human use.
Research-Only Disclaimer: All products are intended for laboratory and research purposes only. Not for human or animal consumption. These statements have not been evaluated by the FDA.