Last Updated: April 14, 2026
Regenerative peptide research has expanded rapidly over the past decade, with two molecules attracting particular attention in the preclinical literature: BPC-157, a pentadecapeptide derived from a protective sequence found in human gastric juice, and GHK-Cu, a copper-bound tripeptide naturally present in human plasma. Though often grouped together under the umbrella of “regenerative peptides,” the two compounds operate on very different axes. BPC-157 is most frequently studied in systemic soft-tissue contexts, where published studies suggest it modulates angiogenesis and cytoprotective signaling across the gut, tendon, and vascular compartments. GHK-Cu, by contrast, is primarily investigated in dermal and extracellular matrix remodeling models, where copper delivery appears central to fibroblast activation and collagen turnover. This guide compares BPC-157 vs GHK-Cu across mechanism, tissue focus, and research applications, with the goal of helping laboratories select the appropriate tool compound for their research protocols.
Quick Comparison: BPC-157 vs GHK-Cu
| Attribute | BPC-157 | GHK-Cu |
|---|---|---|
| Peptide class | Pentadecapeptide (15 amino acids) | Copper-bound tripeptide (3 amino acids) |
| Sequence | GEPPPGKPADDAGLV | Glycyl-L-histidyl-L-lysine + Cu(II) |
| Natural origin | Fragment of human gastric juice protein | Endogenous plasma tripeptide |
| Primary research focus | Systemic soft-tissue and gut models | Dermal, collagen, and matrix models |
| Key pathways | VEGF-A, nitric oxide (NO), FAK-paxillin | Collagen I/III synthesis, GRP78, TGF-β |
| Metal cofactor | None | Cu(II) — integral to activity |
| Reconstitution | Bacteriostatic water, refrigerated | Bacteriostatic water, protected from light |
| Stability in solution | Good (7-14 days refrigerated) | Moderate (light/oxidation sensitive) |
What Are Regenerative Peptides?
Regenerative peptides are short amino acid sequences investigated for their role in modulating tissue repair processes in preclinical models. Unlike growth factors, which are large signaling proteins, peptides typically comprise 3 to 40 residues and often mimic bioactive fragments of larger endogenous molecules. Published research indicates that regenerative peptides act through a combination of receptor binding, metal ion delivery, and modulation of downstream transcription factors that govern cell migration, proliferation, and extracellular matrix deposition.
Within this category, two broad mechanistic subclasses are frequently compared. The first includes peptides like BPC-157, which appear to exert systemic effects via vascular and cytoprotective pathways regardless of delivery site. The second includes peptides such as GHK-Cu that are studied primarily in dermal and matrix remodeling contexts, where localized fibroblast and keratinocyte activity dominates the observed response. Understanding which category a peptide belongs to is critical when designing laboratory use protocols, because the tissue system being modeled determines which biomarkers, readouts, and time courses make sense. For a broader orientation to this category, see the healing peptides research pillar.
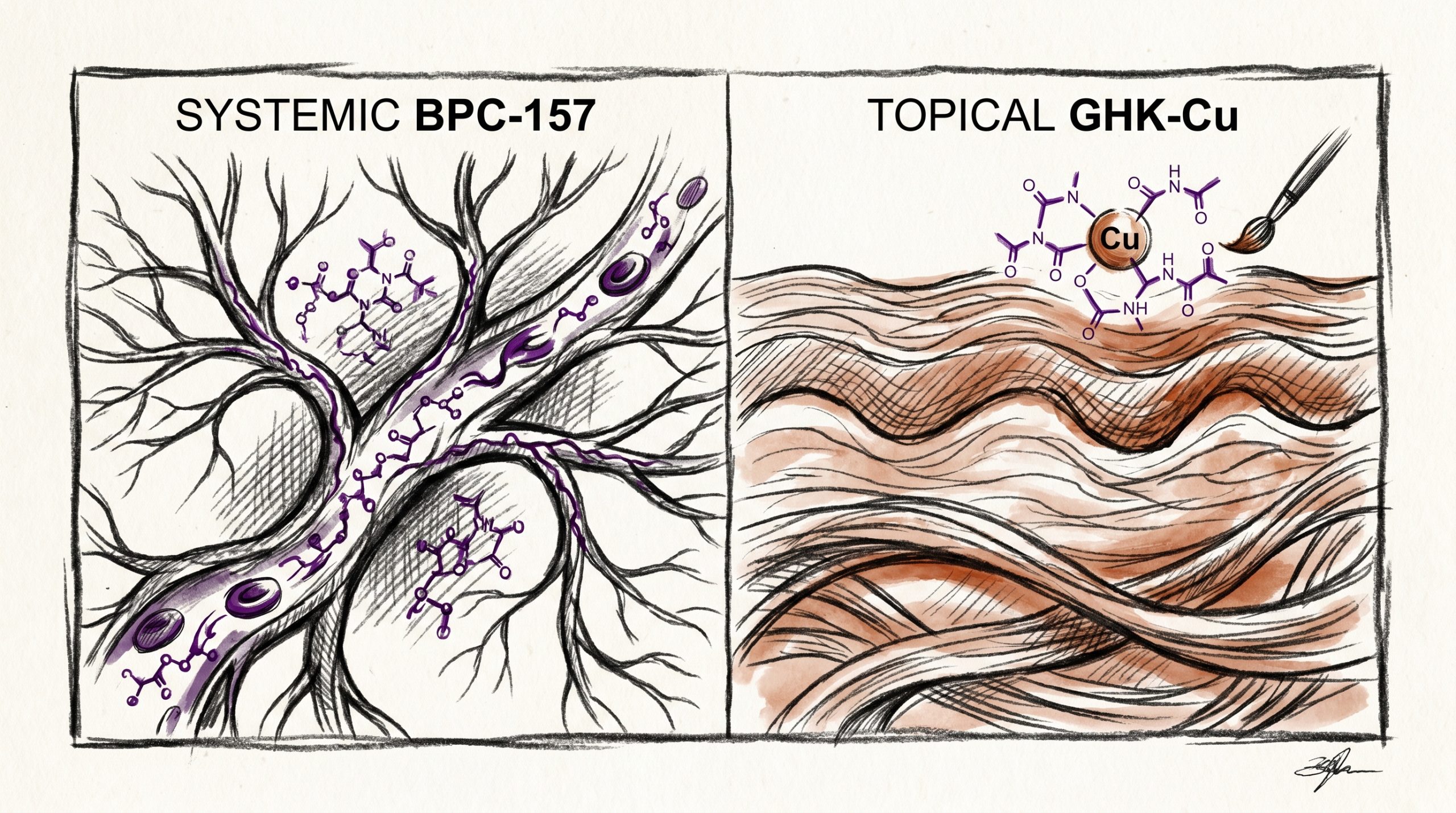
How Does BPC-157 Support Systemic Tissue Research?
BPC-157, or Body Protection Compound-157, is a stable synthetic pentadecapeptide whose parent sequence was isolated from a gastric juice protein that resists acid denaturation. Its stability profile is unusual; preclinical research indicates the molecule remains active in gastric conditions that would rapidly degrade most peptides, which is part of why it has been studied in such a wide range of systemic models. BPC-157 (10mg) is commonly reconstituted with bacteriostatic water for laboratory use at micromolar concentrations.
Published studies suggest that BPC-157’s core mechanism centers on two interlinked pathways: vascular endothelial growth factor (VEGF-A) signaling and nitric oxide (NO) modulation. In preclinical soft-tissue models — tendon, muscle, ligament, and gut epithelium — administration has been associated with upregulation of VEGFR2, accelerated angiogenic sprouting, and preservation of microvascular integrity following ischemic or chemical insult. Research protocols examining Achilles tendon transection in rodent models have documented enhanced fibroblast recruitment and organized collagen fibril alignment compared with saline controls. Additional preclinical research indicates that BPC-157 interacts with the nitric oxide system in a context-dependent manner, buffering both hypotensive and hypertensive perturbations rather than forcing the system in one direction.
A third mechanistic theme emerging in the literature involves FAK-paxillin signaling in focal adhesion assembly, which is relevant to cell migration assays in vitro. For laboratories modeling systemic soft-tissue repair — particularly gut, tendon, or vascular systems — BPC-157 represents one of the most extensively characterized research tool compounds available.
How Does GHK-Cu Modulate Dermal and Collagen Remodeling?
GHK-Cu is a naturally occurring tripeptide composed of glycine, histidine, and lysine, bound to a copper(II) ion with exceptionally high affinity. Plasma concentrations of GHK decline substantially with age in humans, and this decline correlates in preclinical research with reduced dermal fibroblast activity and slower extracellular matrix turnover. GHK-Cu (50mg) is typically reconstituted in bacteriostatic water and protected from light during storage, as the copper-peptide complex is sensitive to oxidation.
Unlike BPC-157, GHK-Cu’s activity is inseparable from its copper cofactor. Published studies suggest the peptide functions as a copper chaperone, delivering Cu(II) to enzymes involved in collagen cross-linking (notably lysyl oxidase) and superoxide dismutase activity. This delivery mechanism is why the literature treats GHK and GHK-Cu as distinct entities; the apo-peptide without copper shows minimal activity in most dermal research assays.
In dermal fibroblast cultures, preclinical research indicates GHK-Cu upregulates type I and type III collagen synthesis, elastin, glycosaminoglycans, and decorin. Keratinocyte models show enhanced migration and integrin expression. GHK-Cu has also been studied for its modulation of GRP78 and antioxidant response element (ARE) transcription, positioning it as a candidate for research into oxidative stress in skin and barrier tissue models. Gene expression studies using the Broad Institute Connectivity Map have indicated GHK signatures resembling those of tissue-remodeling and anti-fibrotic transcriptional programs, which has broadened its use in research protocols beyond purely cosmetic dermal contexts.
Where Does Each Excel in Research?
Selecting between BPC-157 and GHK-Cu depends almost entirely on the tissue system under investigation. A useful decision framework:
- Systemic soft-tissue or gut models: BPC-157 is the better-characterized choice. Research protocols examining tendon, muscle, gastrointestinal epithelium, or vascular endothelium have the deepest published literature base for BPC-157.
- Dermal, wound-bed, or collagen remodeling models: GHK-Cu is the canonical research compound. Fibroblast proliferation assays, ex vivo skin explant studies, and matrix synthesis readouts consistently feature GHK-Cu as a reference standard.
- Angiogenesis-focused research: Both are relevant, but the mechanism differs. BPC-157 acts primarily through VEGF-A/VEGFR2 induction, while GHK-Cu modulates angiogenesis via copper-dependent endothelial signaling and TGF-β pathways.
- Oxidative stress and antioxidant enzyme models: GHK-Cu has a clearer link to superoxide dismutase and ARE pathways due to its copper delivery role.
- Cytoprotection under chemical insult: BPC-157 has a larger preclinical footprint, particularly in NSAID-induced gut lesion models.
In practice, research groups working across multiple tissue compartments often maintain both compounds in their inventory and select based on the specific model being run that week. They address different biology, and mixing up their use cases can lead to ambiguous readouts.
Why Are They Combined in GLOW/KLOW Research Stacks?
A number of research-stack formulations combine GHK-Cu with other peptides to probe synergistic effects across tissue compartments. Two of the most studied are GLOW and KLOW, both of which pair GHK-Cu with BPC-157 and additional regenerative peptides.
- GLOW Blend (70mg) typically combines GHK-Cu, BPC-157, and TB-500 10mg (thymosin beta-4 fragment). The rationale in preclinical research is that GHK-Cu drives dermal matrix remodeling, BPC-157 provides systemic vascular and cytoprotective support, and TB-500 contributes to cell migration and actin sequestration. Together they span three complementary axes of tissue repair research.
- KLOW Blend (80mg) adds KPV (a tripeptide derived from alpha-MSH with anti-inflammatory signaling in preclinical models) to the GLOW formula. Research protocols investigating combined inflammation, vascular, and matrix endpoints have used KLOW as a multi-axis probe.
The synergy rationale rests on the observation that tissue repair is never driven by a single pathway. Preclinical research indicates that angiogenesis, matrix deposition, inflammation resolution, and cell migration proceed in parallel with tight crosstalk. Stacking peptides that each target a different node of this network allows researchers to probe whether combined signaling produces additive, synergistic, or antagonistic effects compared with single-peptide protocols. Published studies on any specific blend formulation remain limited, so most stack research is exploratory and hypothesis-generating rather than confirmatory.
What Does Published Research Show?
Both peptides have substantial PubMed footprints, though the depth differs by tissue system.
BPC-157 published research includes Sikiric et al. (multiple reviews on PubMed documenting systemic cytoprotection and VEGF pathway modulation in rodent models), Chang et al. on Achilles tendon transection recovery (PubMed ID 21030672), and Huang et al. on nitric oxide system buffering (PubMed literature on BPC-157 and NO-system). The majority of BPC-157 literature is preclinical; human clinical data remain limited and are not the basis for research protocol design.
GHK-Cu published research is anchored by the work of Pickart and Margolina, whose review “Regenerative and Protective Actions of the GHK-Cu Peptide” (PubMed ID 29581963) synthesizes decades of dermal and matrix research. Additional PubMed-indexed studies document GHK-Cu’s effects on collagen and decorin expression in fibroblasts, antioxidant enzyme upregulation, and gene expression signatures consistent with tissue remodeling programs.
For laboratory use, the practical takeaway is that both compounds are supported by peer-reviewed preclinical literature in their respective tissue domains, and research protocols should cite the specific studies most relevant to the model being run.
Frequently Asked Questions
Can BPC-157 and GHK-Cu be combined in research protocols?
Preclinical research indicates that BPC-157 and GHK-Cu operate through largely non-overlapping pathways, which is the mechanistic basis for stacking them in research blends such as GLOW and KLOW. BPC-157 modulates systemic VEGF-A and nitric oxide signaling, while GHK-Cu delivers copper to matrix-remodeling enzymes and activates dermal fibroblasts. Because the two compounds target different nodes in the tissue repair network, published studies suggest that combined use in laboratory protocols does not produce direct receptor competition. However, formal pharmacokinetic interaction studies in preclinical models remain limited, so research groups typically run single-peptide arms alongside any combination arm to isolate effects. All combination work should be conducted under laboratory use conditions with appropriate controls. These compounds are intended for laboratory and research purposes only.
Why is copper important in GHK-Cu?
Copper is integral to GHK-Cu’s activity because the peptide functions as a copper chaperone in preclinical research models. The tripeptide backbone binds Cu(II) with exceptionally high affinity and delivers the ion to enzymes that depend on copper for their function, most notably lysyl oxidase (involved in collagen cross-linking) and superoxide dismutase (an antioxidant enzyme). Published studies indicate that GHK without copper — the apo-peptide — shows substantially reduced activity in most dermal and matrix assays, which is why the copper-bound form is the research standard. This also explains why GHK-Cu must be protected from light and stored carefully, as oxidation of the copper center can compromise the complex. In contrast, BPC-157 has no metal cofactor and operates through peptide-receptor interactions alone.
Which peptide is better for wound healing studies?
The answer depends on the wound model. For dermal wound-bed research — full-thickness skin wounds, burn models, or diabetic wound research in rodents — GHK-Cu has the larger published footprint, because its mechanism directly targets dermal fibroblasts, keratinocyte migration, and collagen remodeling in the wound environment. For systemic tissue repair research — tendon transection, gut mucosal lesion recovery, or muscle crush models — BPC-157 is more extensively characterized in preclinical research. Some laboratories combine both in wound models to probe whether systemic vascular support (BPC-157) complements local matrix deposition (GHK-Cu). Neither compound is approved for human use, and all wound research should be conducted in appropriate animal or in vitro models under research protocols. Results in preclinical models do not imply equivalent effects in humans.
How are BPC-157 and GHK-Cu reconstituted for research?
Both peptides are typically supplied as lyophilized powder and reconstituted for laboratory use with bacteriostatic water. For BPC-157, a common research concentration is reached by adding 2 to 5 mL of bacteriostatic water per 10 mg vial, depending on the desired stock concentration for the assay. GHK-Cu reconstitution follows a similar process, though the solution has a characteristic blue tint from the copper complex and should be protected from light during storage. Reconstituted BPC-157 is generally stable for 7 to 14 days refrigerated, while GHK-Cu solutions are more oxidation-sensitive and often used within a shorter window. All reconstitution should be performed in a clean laboratory environment with sterile technique, and all aliquots should be labeled clearly. These products are for laboratory and research purposes only.
Do BPC-157 and GHK-Cu affect the same signaling pathways?
No — preclinical research indicates the two peptides act on largely distinct signaling networks. BPC-157 is most consistently linked to VEGF-A/VEGFR2 angiogenic signaling, nitric oxide synthase modulation, and FAK-paxillin focal adhesion dynamics. Published studies also suggest activity on the 5-HT and dopaminergic systems in certain preclinical central nervous system models, though this is less well characterized. GHK-Cu, by contrast, operates primarily through copper-dependent enzyme activation (lysyl oxidase, superoxide dismutase), collagen and decorin gene expression, and GRP78-mediated stress response pathways. There is modest overlap in downstream endpoints — both can influence angiogenesis and matrix deposition — but the upstream mechanisms are mechanistically distinct, which is the basis for stacking them in research protocols.
Are there documented concerns with long-term research use?
For laboratory use, the primary considerations are compound purity, solution stability, and assay reproducibility rather than extrapolations to human use. Preclinical research on BPC-157 has reported consistent safety margins in rodent models across the doses tested, though long-term multi-generational studies remain limited in the published literature. GHK-Cu carries the additional consideration of copper loading; at high concentrations in vitro, copper itself can become cytotoxic, so dose-response curves should always be characterized in each specific cell line or tissue model being used. Neither peptide has FDA approval for human use, and the statements in this guide have not been evaluated by the FDA. All research should be conducted under appropriate laboratory oversight, and results in preclinical models should not be interpreted as predictive of human outcomes.
Related research pillar: Healing Peptides Research Guide
All products are intended for laboratory and research purposes only. Not for human or animal consumption. These statements have not been evaluated by the FDA.