Last Updated: May 1, 2026 | v1.0
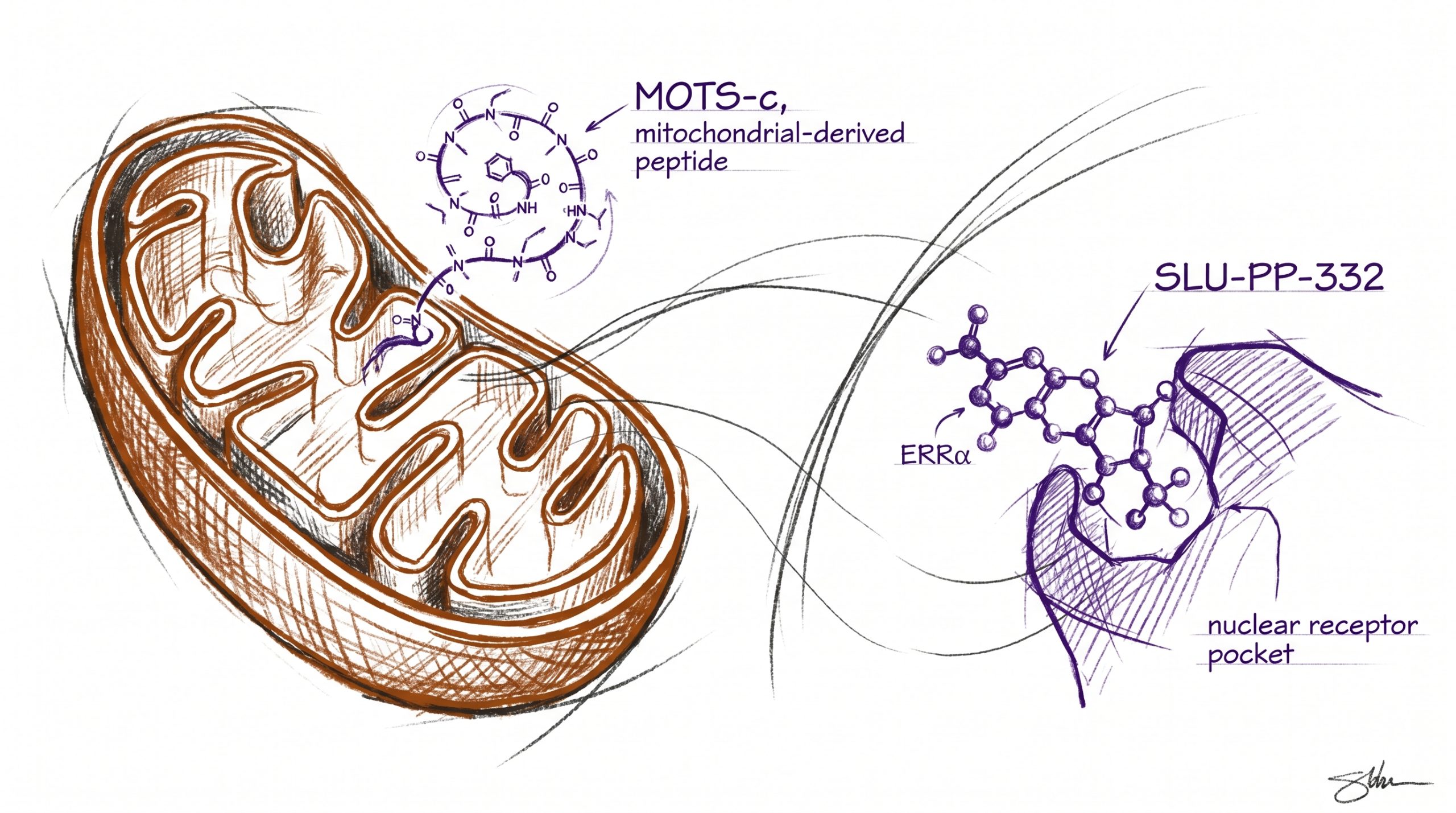
MOTS-c and SLU-PP-332 are two research molecules investigated for their exercise-mimetic activity in preclinical metabolic and longevity studies, but they belong to entirely different chemical classes and engage opposite ends of cellular bioenergetics. MOTS-c is a 16-amino-acid mitochondrial-derived peptide encoded within the 12S rRNA region of mitochondrial DNA, identified in 2015 and studied for its role in AMPK activation, glucose homeostasis, and skeletal muscle metabolism. SLU-PP-332 is a small-molecule synthetic agonist of estrogen-related receptor alpha (ERRα), a nuclear receptor that drives mitochondrial biogenesis transcription programs. Although both are described in published literature as “exercise mimetics,” they operate at different layers of the bioenergetic stack: MOTS-c at the cytosolic AMPK signaling level, SLU-PP-332 at the nuclear-receptor transcription level. This guide compares the two side by side. All products and information are provided for laboratory and research purposes only.
Quick Comparison: MOTS-c vs SLU-PP-332
| Attribute | MOTS-c | SLU-PP-332 |
|---|---|---|
| Chemical class | Peptide (16 amino acids) | Small-molecule synthetic ligand |
| Origin | Encoded in 12S rRNA region of mitochondrial DNA | Designed at Saint Louis University; published 2023 |
| Molecular weight | ~2,174 Da | ~395 Da |
| Primary target | AMPK signaling axis (indirect activation) | Estrogen-Related Receptor alpha (ERRα) — pan-ERR agonist |
| Site of action | Cytosol and nucleus; signals to AMPK and downstream metabolic genes | Nuclear receptor; binds ERR ligand-binding domain to drive transcription |
| Reported preclinical effects | Improved glucose tolerance, insulin sensitivity, AMPK activation, weight regulation in rodent models | Increased exercise endurance, mitochondrial biogenesis, fat oxidation in mice without exercise |
| Route in research | Parenteral (intraperitoneal, subcutaneous in published rodent studies) | Orally bioavailable in published rodent models |
| Literature maturity | Published since 2015; growing multi-laboratory corpus | Emerging; first major publication 2023 (Nature) |
What Is an Exercise-Mimetic in Research Contexts?
The term “exercise mimetic” describes molecules studied for their ability to recapitulate, in part or in whole, the metabolic adaptations that physical exercise produces in skeletal muscle, adipose tissue, liver, and the cardiovascular system. Exercise produces a coordinated cellular response that includes AMPK activation in response to a rising AMP:ATP ratio, mitochondrial biogenesis driven by PGC-1α and the estrogen-related receptor (ERR) family, glucose-uptake increases in muscle, and shifts in substrate utilization toward fatty-acid oxidation. A research molecule that activates one or more of these pathways in the absence of physical contraction is, by working definition, an exercise mimetic. The category is broad and includes well-known compounds such as AICAR (an AMPK activator) and PPAR-delta agonists. MOTS-c and SLU-PP-332 represent two distinct entry points into this signaling network: peptide-driven AMPK activation versus small-molecule nuclear receptor agonism.
Importantly, no exercise mimetic in published preclinical research fully reproduces the integrated, multi-system response of physical exercise. Each candidate engages a subset of the pathways and tissues that exercise activates. Investigators studying exercise-mimetic candidates select among them based on which subset of the response is most relevant to the research question — glucose homeostasis, mitochondrial respiratory capacity, endurance performance, body composition, or longevity-associated transcriptional programs. The MOTS-c versus SLU-PP-332 comparison is illustrative because the two compounds engage non-overlapping primary mechanisms and produce overlapping but non-identical phenotypes in published rodent studies.
How Does MOTS-c Work as a Mitochondrial-Derived Peptide?
MOTS-c (Mitochondrial Open Reading frame of the Twelve S rRNA type-c) is a 16-amino-acid peptide encoded by a small open reading frame within the 12S ribosomal RNA region of mitochondrial DNA. Discovered in 2015 by Lee and colleagues at the University of Southern California (Cell Metabolism, 2015), MOTS-c is one of the first characterized members of an emerging class of “mitochondrial-derived peptides” that cells secrete in response to metabolic stress. The 16-residue sequence (MRWQEMGYIFYPRKLR) localizes initially to mitochondria but is also found in plasma and the nucleus in published studies, where it appears to translocate during metabolic stress to influence transcriptional programs. Researchers studying mitochondrial signaling can source MOTS-c from Peptideware with batch-specific HPLC verification.
Mechanistically, published preclinical studies have characterized MOTS-c as an AMPK pathway activator that improves glucose homeostasis and insulin sensitivity in rodent models. The Cell Metabolism 2015 paper demonstrated that MOTS-c administration to high-fat-diet mice improved insulin sensitivity, reduced adiposity, and activated AMPK signaling in skeletal muscle. Subsequent preclinical work has extended these observations to include effects on metabolic flexibility, mitochondrial function in skeletal muscle, and potential roles in age-associated metabolic decline. MOTS-c levels in human plasma have been reported to decline with age in observational studies, raising hypotheses that endogenous MOTS-c contributes to metabolic homeostasis and that age-related decline may parallel insulin-sensitivity decline. These hypotheses remain under active investigation in published literature, and translation from rodent models to human metabolic biology has not been established. The MOTS-c with DAC variant supplied for research use is a long-acting analog modified with a Drug Affinity Complex (DAC) extension to extend half-life in preclinical pharmacokinetic studies.
How Does SLU-PP-332 Work as an ERRα Agonist?
SLU-PP-332 is a small-molecule synthetic agonist of the estrogen-related receptor (ERR) family of nuclear receptors, with primary activity at ERRα and notable activity at ERRβ and ERRγ. It was characterized in a 2023 publication in Nature describing the design, in vivo testing, and metabolic effects of the molecule in mice (Billon et al., Nature, 2023). The estrogen-related receptors are constitutively active orphan nuclear receptors that drive transcription of mitochondrial biogenesis genes including PGC-1α targets, oxidative phosphorylation components, and fatty-acid oxidation enzymes. SLU-PP-332 binds the ligand-binding domain of ERRα and stabilizes the active conformation, increasing its transcriptional activity in cellular and animal models. Researchers studying ERR-driven metabolic biology can source SLU-PP-332 from Peptideware, supplied as a 5 mg lyophilized preparation for preclinical research.
The most striking finding from the 2023 Nature publication was that orally administered SLU-PP-332 increased running endurance in sedentary mice by approximately 70% relative to vehicle controls in treadmill assays, in the absence of exercise training. The compound also produced reductions in body weight and fat mass in diet-induced-obesity mouse models and increased oxygen consumption consistent with elevated mitochondrial respiratory capacity. Mechanistically, the published transcriptional analysis showed that SLU-PP-332 treatment activated ERRα-driven gene programs in skeletal muscle, including upregulation of oxidative phosphorylation components and fatty-acid oxidation enzymes. These findings positioned SLU-PP-332 as a leading research tool for probing ERRα biology and exercise-mimetic transcriptional programs. Importantly, the clinical-translation status remains open: SLU-PP-332 itself is a research compound, not a drug candidate at the time of publication, and significant additional preclinical work is required before any clinical investigation could proceed under regulatory oversight. The published literature on SLU-PP-332 is still small, with the 2023 Nature paper as the foundational reference and subsequent mechanistic studies emerging.
How Do MOTS-c and SLU-PP-332 Compare in Published Research?
The published research bases for these two molecules differ substantially in size, depth, and breadth. MOTS-c has accumulated nearly a decade of preclinical literature since its 2015 discovery, with multi-laboratory studies across metabolic, cardiovascular, and aging research domains. The MOTS-c corpus includes mechanistic studies of AMPK activation, biodistribution and pharmacokinetic analyses, observational human-plasma studies in aging cohorts, and exploratory studies in cardiac and skeletal muscle biology. SLU-PP-332’s literature is much younger; the 2023 Nature publication established the molecule as a tool, and subsequent published studies are still emerging. For investigators selecting between the two, the maturity of the literature is a meaningful consideration: MOTS-c has more published replication and broader mechanistic characterization, while SLU-PP-332 represents a newer and more sharply defined transcriptional intervention.
| Compound | Primary research area | Representative preclinical findings | Literature characteristics |
|---|---|---|---|
| MOTS-c | Glucose homeostasis, insulin sensitivity, skeletal muscle metabolism, aging | Improved glucose tolerance and AMPK activation in HFD mice (Cell Metabolism 2015); plasma decline with age in human observational data | Multi-laboratory corpus since 2015; mechanistic and observational studies |
| SLU-PP-332 | Mitochondrial biogenesis, exercise endurance, oxidative phosphorylation transcription | ~70% increase in treadmill endurance in sedentary mice; reduced body weight in DIO mice (Nature 2023) | Emerging; foundational 2023 Nature publication, mechanistic follow-up in progress |
A second consideration is the layer of the metabolic-signaling hierarchy each compound engages. MOTS-c activates AMPK, a cytosolic energy-status sensor that phosphorylates downstream targets including ACC, mTORC1 components, and PGC-1α. SLU-PP-332 acts directly at the nuclear-receptor transcriptional level, binding ERRα to drive mitochondrial biogenesis gene programs that PGC-1α also coordinates. The two interventions therefore converge on overlapping downstream programs (mitochondrial biogenesis, fatty-acid oxidation) but enter the network at different upstream nodes. This has implications for combination research: in principle, parallel activation of cytosolic energy-sensing (MOTS-c → AMPK) and nuclear transcription (SLU-PP-332 → ERRα) could produce additive effects on mitochondrial respiratory capacity, but published combination data are not yet available. The pillar guide on longevity peptides places both compounds in the broader metabolic-aging research landscape alongside NAD+ precursors and SS-31.
Which Compound Should Researchers Choose?
Selection between MOTS-c and SLU-PP-332 depends primarily on the research question and the layer of the metabolic-signaling hierarchy under investigation. For investigators studying glucose homeostasis, insulin sensitivity, AMPK pathway activation, or mitochondrial-derived peptide biology in aging contexts, MOTS-c is the appropriate choice. Its peptide nature and the substantial published literature on its metabolic effects make it well-suited to mechanistic studies of insulin signaling, glucose-uptake assays in muscle and adipose preparations, and observational designs that examine endogenous MOTS-c in metabolic disease and aging research. The MOTS-c with DAC long-acting variant is appropriate when extended pharmacokinetic exposure is needed for chronic-administration protocols.
For investigators studying ERR family transcription, mitochondrial biogenesis at the nuclear-receptor level, exercise endurance phenotypes, or oral-route small-molecule interventions in metabolic and longevity research, SLU-PP-332 is the appropriate choice. Its small-molecule nature makes it suitable for cellular reporter assays of ERR activity, in vivo oral-administration protocols in rodent models, and transcriptional studies of mitochondrial gene programs. Investigators should be aware that the published SLU-PP-332 literature is still small, and reference data for many specific tissue contexts and disease models do not yet exist; new preclinical work in this space is breaking ground rather than confirming established findings. For a broader survey of research peptides and metabolic compounds, the Peptideware reference guide provides cross-compound context.
How Are These Compounds Reconstituted and Stored?
MOTS-c is supplied as a lyophilized powder in sealed vials and should be stored at -20 degrees Celsius for long-term stability. For research use, reconstitution with pharmaceutical-grade bacteriostatic water containing 0.9% benzyl alcohol is the standard protocol; the benzyl alcohol preservative supports multi-use vials across a typical 28-day reconstituted window at 2-8 degrees Celsius refrigeration. Reconstituted MOTS-c should not be frozen, because ice crystal formation damages peptide bonds and degrades molecular integrity. Researchers should label each reconstituted vial with the date of reconstitution, concentration, and bacteriostatic water volume, and gently swirl rather than shake during reconstitution to preserve structural fidelity. Peptideware supplies bacteriostatic water in compatible 3 mL and 10 mL formats.
SLU-PP-332 is a small-molecule research chemical and is supplied as a lyophilized solid. Storage at -20 degrees Celsius in sealed vials is recommended for long-term stability. Reconstitution depends on the intended assay: for in vitro work, dimethyl sulfoxide (DMSO) is the most common vehicle in published cellular studies, with appropriate dilution into assay media. For in vivo preclinical research, the published Nature 2023 protocol used oral gavage with vehicle preparations described in the paper’s supplementary methods. Researchers should consult the primary literature for vehicle composition and dosing parameters specific to their target endpoint. Unlike MOTS-c, SLU-PP-332 does not require bacteriostatic water; the appropriate vehicle is determined by the assay format and route of administration.
Frequently Asked Questions
Is MOTS-c the same molecule as MOTS-c with DAC?
MOTS-c with DAC is a long-acting analog of native MOTS-c that has been modified with a Drug Affinity Complex (DAC) extension to extend the molecule’s half-life in preclinical pharmacokinetic studies. The native 16-residue MOTS-c sequence is preserved as the active core, with the DAC moiety attached to alter clearance and biodistribution properties. In rodent research, the DAC-modified variant is studied for chronic-administration protocols that benefit from sustained exposure without daily injections. The pharmacodynamic profile is broadly similar to native MOTS-c at the AMPK pathway level, but the pharmacokinetics differ. Investigators should select the variant that matches the kinetic profile required by the specific research protocol and consult the primary literature for half-life and exposure parameters in their model system.
What is the functional difference between AMPK activation and ERRα agonism?
AMPK and ERRα operate at different layers of the cellular metabolic-signaling network. AMPK is a heterotrimeric serine/threonine kinase activated by elevated AMP:ATP ratios, which acts as a cytosolic energy-status sensor. When activated, AMPK phosphorylates downstream targets including ACC (suppressing fatty-acid synthesis), mTORC1 components (suppressing growth-related anabolism), and transcriptional coactivators including PGC-1α. ERRα, by contrast, is a constitutively active nuclear receptor that binds DNA at ERR response elements and drives transcription of mitochondrial biogenesis and oxidative-phosphorylation gene programs. AMPK activation can secondarily enhance PGC-1α and ERRα-coordinated transcription via PGC-1α phosphorylation, so the two pathways converge functionally even though they enter the signaling network at distinct nodes. MOTS-c targets the upstream AMPK node; SLU-PP-332 targets the downstream nuclear-receptor node directly.
Can MOTS-c and SLU-PP-332 be combined in research protocols?
Combination research using MOTS-c and SLU-PP-332 has not yet been characterized in the published preclinical literature, and no head-to-head or combination data are available at the time of this guide. In principle, parallel activation of cytosolic energy-sensing through MOTS-c-driven AMPK signaling and direct nuclear-receptor transcription through SLU-PP-332-driven ERRα activation could produce additive effects on mitochondrial respiratory capacity and exercise-mimetic transcriptional programs, because the two interventions enter the metabolic network at distinct upstream nodes that converge functionally on overlapping downstream gene programs. Investigators interested in combination research should design parallel-arm studies that compare individual compounds against the combination and should consult the primary literature for each compound’s pharmacokinetics and pharmacodynamics in the specific model system before combining.
Is SLU-PP-332 orally bioavailable in research?
Yes. The 2023 Nature publication describing SLU-PP-332 used oral administration in mouse studies and reported measurable systemic exposure and metabolic effects after oral dosing. As a small molecule of approximately 395 Daltons, SLU-PP-332 is well-suited to oral-route preclinical research, in contrast to peptide compounds that typically require parenteral administration to avoid proteolytic degradation in the gastrointestinal tract. The published vehicle and dosing protocol in the foundational paper provides the reference framework for oral-route research; investigators designing in vivo studies should consult that publication and any subsequent pharmacokinetic literature for vehicle composition, dose-response parameters, and target plasma exposure relevant to their specific endpoint.
Why is MOTS-c described as an aging-associated peptide?
Published observational studies have measured MOTS-c levels in human plasma across age groups and reported declining levels with advancing age, particularly in cohorts with metabolic dysfunction. These observations, combined with preclinical evidence that MOTS-c administration improves glucose homeostasis and insulin sensitivity in rodent models of metabolic stress, have generated working hypotheses in the published literature that endogenous MOTS-c contributes to metabolic-homeostasis maintenance and that age-related decline parallels the development of insulin resistance and metabolic inflexibility associated with aging. These hypotheses remain under active investigation. Translation from rodent models and human observational data to causal claims in human metabolic aging biology has not been established, and researchers should treat the aging-association literature as hypothesis-generating rather than mechanistically definitive at the current state of published evidence.
What purity and verification should researchers require?
Research-grade MOTS-c and SLU-PP-332 should be sourced from suppliers who provide third-party certificate of analysis documentation. For MOTS-c, peptide identity should be verified by HPLC (typically greater than 98% purity) and mass spectrometry confirmation of molecular weight at approximately 2,174 Daltons. For SLU-PP-332, small-molecule identity should be verified by HPLC purity (typically greater than 98%) and mass spectrometry or NMR characterization. Both compounds should be free of detectable endotoxin contamination for in vivo preclinical use. Peptideware supplies both compounds with batch-specific analytical documentation. For detailed evaluation criteria, researchers can consult the Peptideware Peptide Quality: Third-Party Testing guide, which describes purity standards and COA interpretation for preclinical procurement.
Are these compounds approved for human use?
No. MOTS-c and SLU-PP-332 are supplied exclusively for laboratory and research purposes in preclinical model systems. Neither compound is approved by the FDA for human therapeutic use, and neither should be used in any context outside controlled research environments. SLU-PP-332 is an emerging research tool molecule first published in 2023 and has not undergone the preclinical safety and pharmacokinetic characterization required to support clinical investigation. MOTS-c, despite its longer published research history, is similarly a research compound rather than an approved therapeutic. Peptideware’s products are sold with explicit research-use-only documentation, and researchers should ensure that all preclinical protocols comply with applicable institutional and regulatory requirements.
Summary
MOTS-c and SLU-PP-332 are two distinct research molecules studied for exercise-mimetic activity but operating at different layers of the cellular bioenergetic network. MOTS-c is a 16-amino-acid mitochondrial-derived peptide that activates AMPK signaling and improves glucose homeostasis in published rodent models, with a multi-laboratory corpus dating to its 2015 discovery. SLU-PP-332 is a small-molecule ERRα agonist published in Nature in 2023 that drives mitochondrial biogenesis transcription and produced approximately 70% increases in treadmill endurance in sedentary mice. Researchers selecting between them should match the compound’s mechanistic layer (cytosolic AMPK versus nuclear ERR transcription) to the research question, and should consult primary literature for pharmacokinetic and pharmacodynamic parameters before protocol design. Both compounds are research tools only and have not been approved for human therapeutic use.
Research Disclaimer: All products are intended for laboratory and research purposes only. Not for human or animal consumption. These statements have not been evaluated by the FDA.