Last Updated: May 8, 2026 | v1.0
NAD+ and SLU-PP-332 are two research compounds investigated in published metabolic and longevity studies, but they belong to fundamentally different molecular categories and engage cellular bioenergetics through opposite roles. NAD+ (nicotinamide adenine dinucleotide) is an endogenous redox coenzyme present in every living cell, consumed and regenerated continuously in central metabolism, and required as an obligate substrate by sirtuin deacetylases and PARP enzymes. SLU-PP-332 is a synthetic small-molecule agonist of estrogen-related receptor alpha (ERRα), a nuclear receptor that drives transcription of mitochondrial biogenesis gene programs. NAD+ is a substrate; SLU-PP-332 is a ligand. Their published research literatures, mechanisms, and protocol contexts differ accordingly. This guide compares the two compounds for researchers selecting between them in metabolic and longevity studies. All products and information are provided for laboratory and research purposes only.
Quick Comparison: NAD+ vs SLU-PP-332
| Attribute | NAD+ | SLU-PP-332 |
|---|---|---|
| Chemical class | Endogenous dinucleotide coenzyme | Small-molecule synthetic ligand |
| Origin | Naturally present in all living cells; supplied as a research-grade preparation | Designed at Saint Louis University; published 2023 |
| Molecular weight | ~663 Da | ~395 Da |
| Functional role | Substrate / cofactor: redox cycling (NAD+/NADH) and sirtuin/PARP enzyme co-substrate | Receptor agonist: binds ERRα ligand-binding domain to drive transcription |
| Primary research targets | Sirtuins (SIRT1–7), PARPs, CD38, redox enzymes, mitochondrial respiration | Estrogen-Related Receptor alpha (with activity at ERRβ and ERRγ) |
| Reported preclinical effects | Sirtuin activity support, mitochondrial respiratory capacity, age-associated NAD+ decline studies | Increased exercise endurance, mitochondrial biogenesis, fat oxidation in mice without exercise |
| Route in research | Parenteral (intraperitoneal, intravenous, subcutaneous in published rodent studies); precursors used orally | Orally bioavailable in published rodent models |
| Literature maturity | Decades-spanning corpus across biochemistry, aging, and metabolism | Emerging; foundational 2023 Nature publication |
What Is NAD+ and Why Does It Matter in Longevity Research?
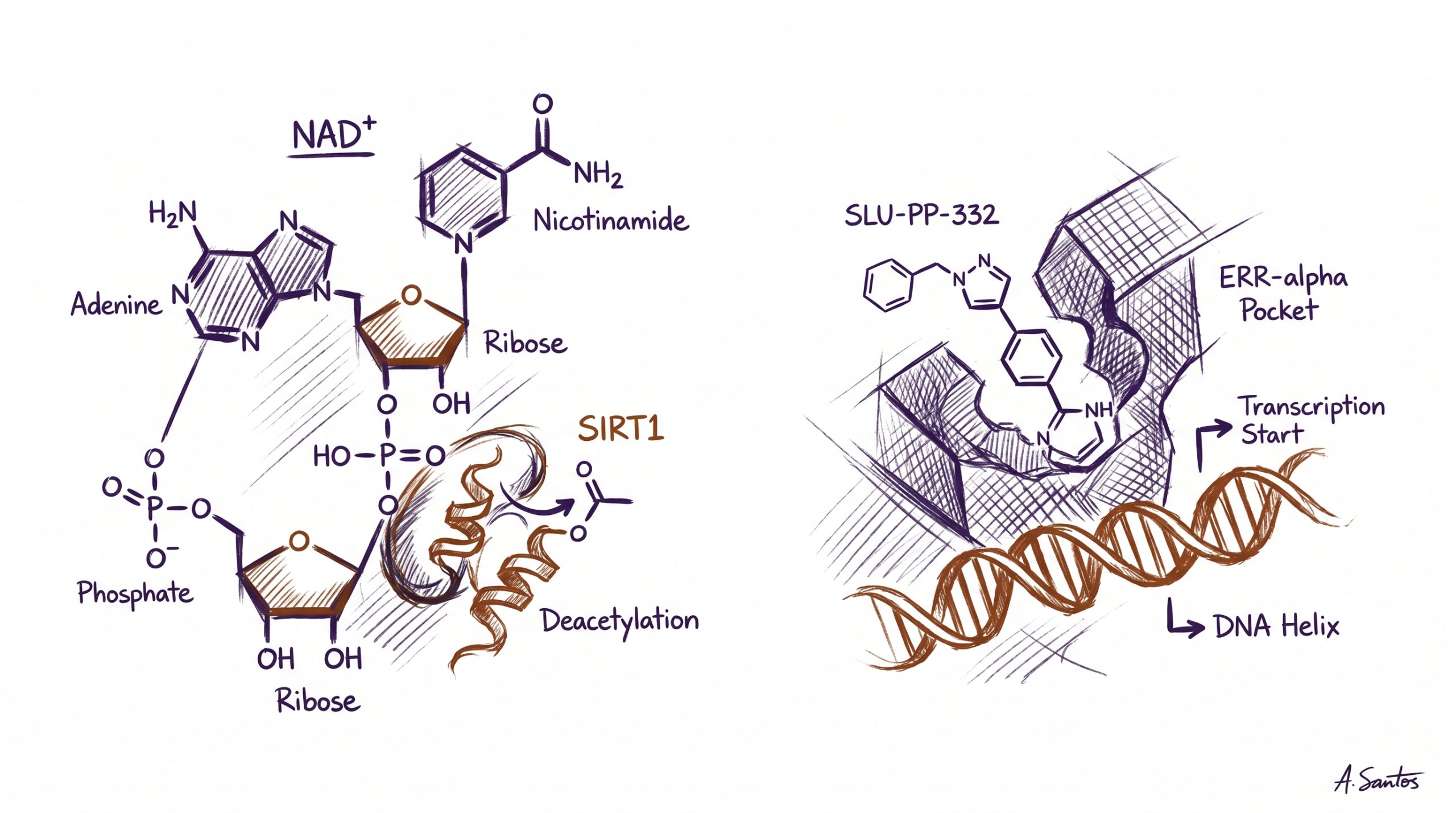
Nicotinamide adenine dinucleotide (NAD+) is a dinucleotide coenzyme composed of an adenine nucleotide and a nicotinamide nucleotide linked through a pyrophosphate bridge. It is one of the most abundant and metabolically central small molecules in cellular biology, present in every living cell and continuously consumed and regenerated through redox cycling between its oxidized form (NAD+) and reduced form (NADH). In glycolysis, the citric-acid cycle, and the electron-transport chain, NAD+ accepts electrons during substrate oxidation and donates them at the inner mitochondrial membrane to drive oxidative phosphorylation. Beyond its redox role, NAD+ is also an obligate co-substrate for several non-redox enzyme families that consume it stoichiometrically — including the sirtuins (SIRT1 through SIRT7), the PARP family of DNA-repair enzymes, and CD38, an ectoenzyme that hydrolyzes NAD+ as part of immune signaling. Researchers studying coenzyme biology can source NAD+ from Peptideware as a 500 mg lyophilized preparation with batch-specific analytical documentation.
The longevity-research interest in NAD+ stems from two converging lines of published evidence. First, multiple observational studies have reported declines in tissue and plasma NAD+ levels with advancing age, with some published cohorts showing reductions of approximately 50% across the human lifespan. Second, sirtuins — the NAD+-consuming deacetylase family — have been characterized as regulators of metabolic, mitochondrial, and stress-response gene programs in published preclinical work, including PGC-1α deacetylation by SIRT1, FOXO-family activation, and mitochondrial-protein deacetylation by SIRT3. The working hypothesis in much of the published longevity literature is that age-associated NAD+ decline limits sirtuin activity, which in turn contributes to age-associated metabolic and mitochondrial deterioration. This hypothesis remains under active investigation; causal claims in human aging biology have not been established, and a substantial portion of the research uses NAD+ precursors (NMN, NR) rather than NAD+ itself. Peptideware’s 500 mg NAD+ preparation supplies the coenzyme directly for in vitro assay use and for preclinical research where the parent compound is preferred to a precursor. The NAD+ Research Guide covers coenzyme biology, sirtuin substrate biochemistry, and protocol considerations in greater depth.
How Does SLU-PP-332 Work as an ERRα Agonist?
SLU-PP-332 is a small-molecule synthetic agonist of the estrogen-related receptor (ERR) family of nuclear receptors, with primary activity at ERRα and additional activity at ERRβ and ERRγ. It was characterized in a 2023 publication in Nature describing the design, in vivo testing, and metabolic effects of the molecule in mice (Billon et al., 2023). The estrogen-related receptors are constitutively active orphan nuclear receptors that drive transcription of mitochondrial biogenesis gene programs, including PGC-1α-coordinated targets, oxidative-phosphorylation components, and fatty-acid oxidation enzymes. SLU-PP-332 binds the ligand-binding domain of ERRα and stabilizes its active conformation, increasing transcriptional output in both cellular and animal models. Investigators studying ERR-driven metabolic biology can source SLU-PP-332 from Peptideware, supplied as a 5 mg lyophilized preparation with analytical documentation. The dedicated SLU-PP-332 Research Guide details ligand pharmacology, dosing context from the published literature, and ERR family biology.
The most striking finding from the 2023 Nature publication was that orally administered SLU-PP-332 increased running endurance in sedentary mice by approximately 70% relative to vehicle controls in treadmill assays, in the absence of any exercise training. The compound also produced reductions in body weight and adiposity in diet-induced-obesity mouse models and increased oxygen consumption consistent with elevated mitochondrial respiratory capacity. Transcriptional analysis showed that SLU-PP-332 treatment activated ERRα-coordinated gene programs in skeletal muscle, including upregulation of oxidative-phosphorylation components and fatty-acid oxidation enzymes. These findings positioned SLU-PP-332 as a leading research tool for probing ERR biology and exercise-mimetic transcriptional programs. The clinical-translation status remains open: SLU-PP-332 is a research compound, not a drug candidate, and substantial additional preclinical work is required before any human investigation could proceed under regulatory oversight. The published literature on SLU-PP-332 is still small; the 2023 Nature paper is the foundational reference, with mechanistic follow-up emerging.
How Do NAD+ and SLU-PP-332 Engage Mitochondrial Biogenesis Differently?
The most important conceptual distinction between these two compounds is the mode in which they interact with cellular biology. NAD+ is a substrate: it is consumed stoichiometrically by sirtuins, PARPs, and CD38, and the rate at which sirtuin-mediated deacetylation can proceed is limited by NAD+ availability. Increasing intracellular NAD+ supports increased flux through sirtuin-dependent reactions but does not itself activate any specific transcription factor. SLU-PP-332 is a ligand: it binds the ERRα receptor, stabilizes the active conformation, and increases the transcriptional output of an already-functional receptor. The two interventions therefore engage cellular regulation at different layers — substrate-supply for an enzyme family on one side, conformational activation of a transcription factor on the other.
Despite this categorical difference, the downstream programs converge on overlapping biology. SIRT1 is one of the most-studied sirtuins, and one of its central preclinical-research substrates is PGC-1α, the transcriptional coactivator that drives mitochondrial biogenesis through cooperation with — among others — the ERR family. When SIRT1 deacetylates PGC-1α (an NAD+-dependent reaction), it enhances PGC-1α coactivator activity at ERR response elements and other PGC-1α-coordinated promoters. SLU-PP-332 enters the same downstream network at the receptor itself, increasing ERRα activity directly. Researchers comparing the two compounds therefore see an example of network convergence: a substrate-driven enzymatic input (NAD+ → SIRT1 → PGC-1α deacetylation → ERRα coactivation) and a direct receptor agonism (SLU-PP-332 → ERRα activation) that ultimately influence overlapping transcriptional outputs. The pillar overview Longevity Peptides: NAD+, SS-31, MOTS-c, and SLU-PP-332 places both compounds in the broader longevity-research context.
| Compound | Mode of action | Representative preclinical findings | Literature characteristics |
|---|---|---|---|
| NAD+ | Endogenous coenzyme; redox carrier and sirtuin/PARP/CD38 substrate | Tissue NAD+ decline with age in observational cohorts; sirtuin-dependent deacetylation of PGC-1α and FOXO substrates in published mechanistic studies | Decades-spanning, multi-laboratory; biochemistry, aging, and metabolism domains |
| SLU-PP-332 | Synthetic agonist of ERRα (with ERRβ/γ activity); stabilizes active receptor conformation | ~70% increase in treadmill endurance in sedentary mice; reduced body weight in DIO mice (Nature 2023) | Emerging; foundational 2023 Nature publication, mechanistic follow-up underway |
Which Compound Should Researchers Choose?
Selection between NAD+ and SLU-PP-332 depends on the research question and the cellular layer under investigation. For investigators studying sirtuin biology, redox cycling, NAD+/NADH ratios, age-associated coenzyme decline, or PARP-mediated DNA-repair biology, NAD+ is the appropriate choice. The 500 mg preparation supports cellular and biochemical assays — sirtuin activity assays, NADase or CD38 enzymology, redox-balance measurements in isolated mitochondria, and supplementation studies in cell culture or rodent models. Investigators studying NAD+ precursor biology (NMN, NR) should source those precursors specifically; the parent compound is the appropriate selection when the research question concerns the coenzyme itself rather than salvage-pathway flux from a precursor. The complementary NAD+ vs SS-31 comparison covers a different mitochondrial-research pairing — coenzyme support versus inner-membrane cardiolipin protection — that is also commonly evaluated together in longevity protocols.
For investigators studying ERR family transcription, mitochondrial biogenesis at the nuclear-receptor level, exercise-mimetic phenotypes, or oral-route small-molecule interventions in metabolic and longevity research, SLU-PP-332 is the appropriate choice. Its small-molecule nature suits cellular reporter assays of ERR activity, in vivo oral-administration protocols in rodent models, and transcriptional studies of mitochondrial gene programs. Investigators should be aware that the SLU-PP-332 corpus is still small, and reference data for many tissue contexts and disease models do not yet exist; new preclinical work is breaking ground rather than confirming established findings. The companion comparison MOTS-c vs SLU-PP-332 contrasts SLU-PP-332 against a peptide-based AMPK input to the same downstream mitochondrial-biogenesis network, providing a third reference point for compound selection.
How Are These Compounds Reconstituted and Stored?
NAD+ is supplied as a lyophilized powder in sealed vials and should be stored at -20 degrees Celsius for long-term stability, with light protection recommended. For research use, reconstitution depends on the assay: in vitro work commonly uses sterile water or buffered saline at concentrations appropriate to the enzymatic or cellular endpoint. NAD+ is unstable in alkaline solutions and at elevated temperatures, and reconstituted stocks should be kept on ice during experimental work and stored at -20 or -80 degrees Celsius for short-term re-use. Investigators preparing intracellular-delivery preparations (for example, liposomal formulations) should consult the relevant protocol literature for vehicle composition. Peptideware supplies bacteriostatic water in 3 mL and 10 mL formats compatible with peptide and small-molecule reconstitution where a buffered preparation is suitable.
SLU-PP-332 is a small-molecule research chemical and is supplied as a lyophilized solid. Storage at -20 degrees Celsius in sealed vials is recommended for long-term stability. Reconstitution depends on the intended assay: for in vitro work, dimethyl sulfoxide (DMSO) is the most common vehicle in published cellular studies, with appropriate dilution into assay media. For in vivo preclinical research, the published 2023 Nature protocol used oral gavage with the vehicle preparation described in the paper’s supplementary methods. Researchers should consult the primary literature for vehicle composition and dosing parameters specific to the target endpoint. Unlike NAD+, SLU-PP-332 does not require aqueous reconstitution; the appropriate vehicle is determined by the assay format and the route of administration.
Frequently Asked Questions
Is NAD+ the same as NMN or NR?
No. NMN (nicotinamide mononucleotide) and NR (nicotinamide riboside) are biosynthetic precursors that cells convert to NAD+ through the salvage pathway. Each precursor enters the salvage pathway at a different step: NR is phosphorylated by NRK enzymes to NMN, and NMN is then adenylated by NMNAT enzymes to NAD+. Researchers studying salvage-pathway flux, oral bioavailability of precursors, or organ-level NAD+ delivery via systemic supplementation typically use NMN or NR. Researchers studying NAD+ biochemistry directly — sirtuin substrate kinetics, redox cycling in isolated mitochondria, or PARP enzymology — work with NAD+ itself. The Peptideware 500 mg NAD+ preparation supplies the parent coenzyme; precursor research requires sourcing NMN or NR separately.
What is the functional difference between sirtuin activation through NAD+ and ERRα activation through SLU-PP-332?
The functional difference is substrate-supply versus receptor agonism. Sirtuins are NAD+-dependent deacetylases whose enzymatic rate is influenced by intracellular NAD+ availability; supplying NAD+ supports flux through sirtuin reactions but does not directly recruit or activate the enzyme above its baseline activity per substrate molecule. ERRα is a constitutively active nuclear receptor whose transcriptional output is increased by ligand binding; SLU-PP-332 occupies the ligand-binding domain and increases ERRα activity at its response elements directly. The two pathways converge functionally because SIRT1 deacetylates PGC-1α, the transcriptional coactivator that cooperates with ERRα at mitochondrial biogenesis genes — but the upstream interventions enter the network at distinct nodes: substrate-supply for an enzyme family in one case, conformational activation of a transcription factor in the other.
Can NAD+ and SLU-PP-332 be combined in research protocols?
Combination research using NAD+ and SLU-PP-332 has not been characterized in published preclinical literature, and no head-to-head or combination data are currently available. In principle, combining a substrate-supply input that supports SIRT1-mediated PGC-1α deacetylation with a direct ERRα ligand could produce additive effects on mitochondrial biogenesis transcription, because the two interventions converge on overlapping gene programs through different mechanistic entry points. Investigators interested in combination research should design parallel-arm studies that compare each compound alone against the combination, control for baseline NAD+ status in the model system, and consult published pharmacokinetic and pharmacodynamic data for each compound before designing dosing protocols. The combination is a hypothesis worth testing rather than an established protocol.
Is SLU-PP-332 orally bioavailable in research?
Yes. The 2023 Nature publication describing SLU-PP-332 used oral administration in mouse studies and reported measurable systemic exposure and metabolic effects after oral dosing. As a small molecule of approximately 395 Daltons, SLU-PP-332 is well-suited to oral-route preclinical research, in contrast to the parent NAD+ coenzyme, which is generally not taken up intact through the gastrointestinal tract and is administered parenterally in published rodent studies (intraperitoneal, intravenous, or subcutaneous). For oral-route research targeting the NAD+ system, investigators typically use precursors (NMN, NR) rather than NAD+ itself.
Why do tissue NAD+ levels decline with age?
Published research on age-associated NAD+ decline has identified contributing factors that include increased CD38 expression in aged tissues (CD38 is a major NAD+-consuming ectoenzyme), reduced biosynthetic capacity through the salvage pathway, and increased PARP activation in response to age-associated DNA-damage accumulation. These factors are not mutually exclusive, and the relative contribution of each varies by tissue, model system, and the published study under consideration. The functional consequence in published work is reduced sirtuin and PARP enzymatic flux, reduced mitochondrial respiratory function in some models, and altered redox balance. Translation from rodent and observational human-cohort data to causal claims in human aging biology has not been established, and the published longevity literature treats NAD+ decline as a hypothesis-generating observation that supports continued investigation rather than as a definitively proven causal mechanism.
What purity and verification should researchers require?
Research-grade NAD+ and SLU-PP-332 should be sourced from suppliers providing third-party certificate of analysis documentation. For NAD+, identity should be verified by HPLC purity (typically greater than 95% as a coenzyme preparation) and mass spectrometry confirmation at approximately 663 Daltons. For SLU-PP-332, small-molecule identity should be verified by HPLC purity (typically greater than 98%) and mass spectrometry or NMR characterization. Both compounds should be free of detectable endotoxin contamination for in vivo preclinical use and should ship with batch-specific analytical documentation. The Peptideware Peptide Quality: Third-Party Testing guide describes purity standards and COA interpretation in detail and applies to both peptide and small-molecule research preparations.
Are these compounds approved for human use?
No. NAD+ and SLU-PP-332 supplied for research are intended exclusively for laboratory and preclinical model-system use. SLU-PP-332 is an emerging research tool molecule first published in 2023 and has not undergone the preclinical safety and pharmacokinetic characterization required to support clinical investigation. NAD+ is an endogenous coenzyme present in every cell, but research preparations are not approved drug products and should not be used outside controlled research contexts. Some clinical-research programs are investigating NAD+ precursors (NMN, NR) under regulatory oversight, but those investigations are distinct from research-grade NAD+ supplied for preclinical use. Peptideware’s products are sold with explicit research-use-only documentation, and researchers should ensure that all preclinical protocols comply with applicable institutional and regulatory requirements.
Summary
NAD+ and SLU-PP-332 are two research compounds investigated in metabolic and longevity contexts but operating through fundamentally different molecular roles. NAD+ is an endogenous redox coenzyme and obligate substrate for sirtuin, PARP, and CD38 enzymes, with a decades-spanning published literature that includes age-associated tissue-level decline and sirtuin-dependent regulation of mitochondrial gene programs through PGC-1α deacetylation. SLU-PP-332 is a synthetic small-molecule agonist of ERRα published in Nature in 2023 that drives mitochondrial biogenesis transcription directly and produced approximately 70% increases in treadmill endurance in sedentary mice. The two pathways converge on overlapping mitochondrial-biogenesis gene programs through distinct upstream entry points — substrate-supply for sirtuin enzymology in the NAD+ case, direct receptor agonism for ERRα in the SLU-PP-332 case. Researchers selecting between them should match the compound’s mechanistic role to the research question, consult primary literature for protocol parameters, and follow third-party COA verification standards. Both compounds are research tools only and have not been approved for human therapeutic use.
Research Disclaimer: All products are intended for laboratory and research purposes only. Not for human or animal consumption. These statements have not been evaluated by the FDA.