Last Updated: April 14, 2026
Nicotinamide adenine dinucleotide (NAD+) is a pyridine coenzyme present in every living cell and central to hundreds of enzymatic reactions governing energy metabolism, genomic stability, and cellular signaling. In contrast with most compounds catalogued on Peptideware, NAD+ is not technically a peptide — it is a dinucleotide built from nicotinamide and adenine linked through two ribose sugars and a pyrophosphate bridge. Researchers study NAD+ because cellular pools decline measurably with age across mammalian tissues, and because published studies suggest that restoring NAD+ availability modulates sirtuin activity, mitochondrial output, and DNA-repair capacity. This NAD+ research guide summarises the coenzyme’s biology, its relationship with sirtuins and PARP enzymes, the framework for aging-related investigations, and the laboratory handling practices applicable to the 500mg vial format offered for research protocols. All material below describes preclinical work; nothing here is medical guidance. For a broader catalogue perspective see our longevity peptides guide.
Quick Facts
- Compound class: Pyridine dinucleotide coenzyme (not a peptide)
- Chemical formula: C21H27N7O14P2 (oxidised form)
- Molecular weight: 663.43 g/mol
- Research format: 500 mg lyophilised powder per vial
- Primary research domains: mitochondrial bioenergetics, sirtuin biology, DNA repair, aging models
- Key partner enzymes: SIRT1, SIRT3, SIRT6, PARP1, CD38
- Storage: -20°C lyophilised; 2-8°C short-term reconstituted
What is NAD+ and why is it classified as a coenzyme rather than a peptide?
NAD+ is a nucleotide-based redox cofactor first isolated by Harden and Young in 1906 during fermentation studies. Structurally it consists of two nucleotides — nicotinamide mononucleotide and adenosine monophosphate — joined by a pyrophosphate linkage, giving it a dinucleotide architecture rather than the amide-linked amino-acid chain that defines a peptide. Peptideware catalogues NAD+ alongside peptide research compounds because investigators studying mitochondrial peptides such as SS-31, MOTS-c, and Humanin frequently examine NAD+ pools in parallel experiments. The oxidised form (NAD+) accepts a hydride ion to become NADH, and this redox cycling is what makes the molecule indispensable for glycolysis, the tricarboxylic acid cycle, and oxidative phosphorylation. Published reviews in Cell Metabolism (Rajman et al., 2018) catalogue the coenzyme’s expanding role beyond redox chemistry into signalling.
How does NAD+ participate in the electron transport chain?
Within the mitochondrial matrix, NADH generated during the TCA cycle donates electrons to Complex I (NADH:ubiquinone oxidoreductase), initiating the proton-pumping cascade that builds the electrochemical gradient across the inner mitochondrial membrane. This gradient drives ATP synthase, producing the bulk of cellular ATP. Preclinical research indicates that the NAD+/NADH ratio is a primary determinant of ETC throughput: when NAD+ availability falls, flux through Complex I slows, reactive oxygen species escape increases, and ATP output declines. A 2016 Science paper by Zhang et al. demonstrated in aged mice that restoring NAD+ precursor availability rescued mitochondrial and stem-cell function in skeletal muscle, reinforcing the coenzyme’s position as a master regulator of bioenergetic capacity in mammalian tissue models used by laboratory researchers.
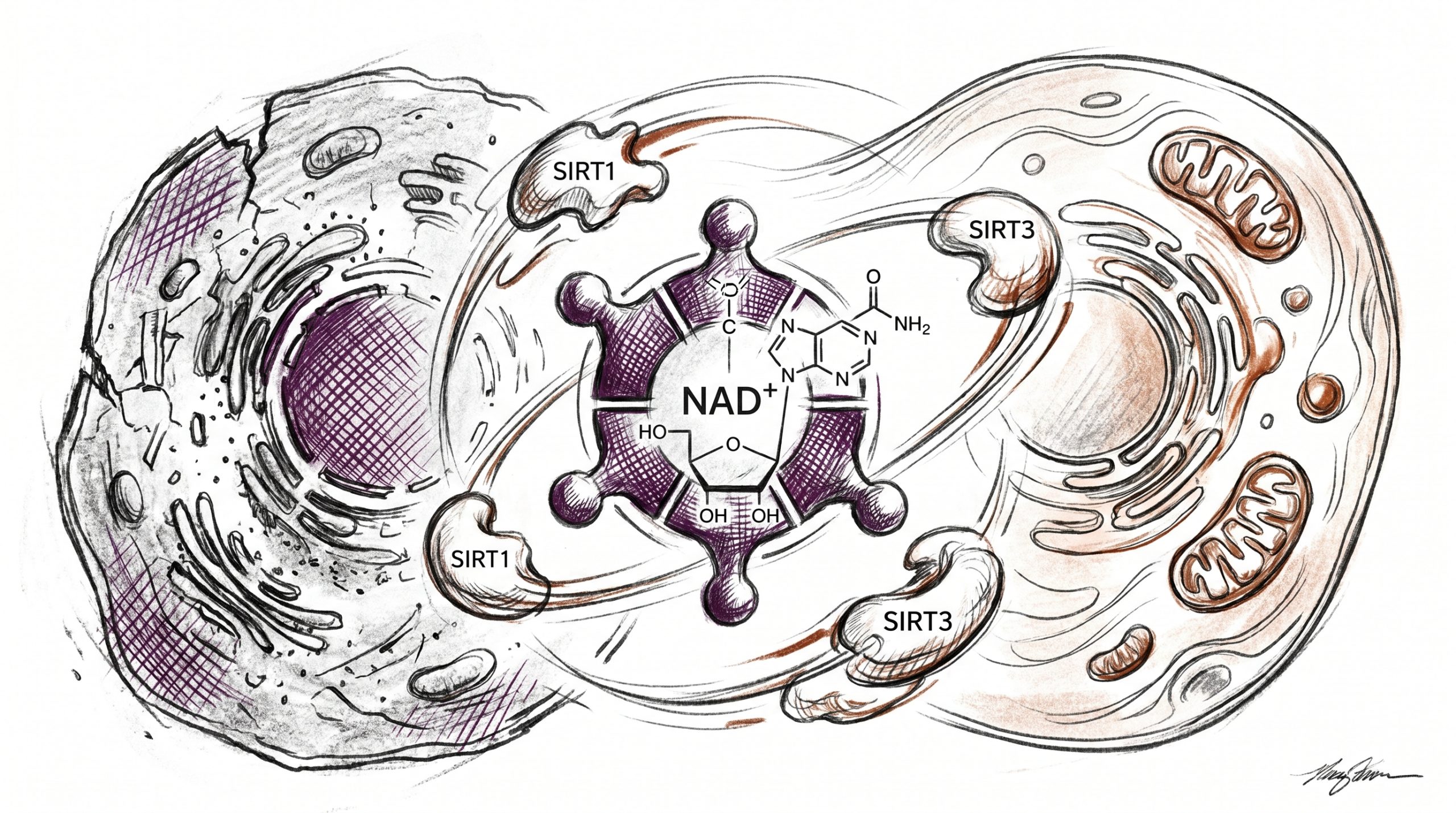
What is the sirtuin pathway and how does NAD+ activate it?
Sirtuins are a family of seven NAD+-dependent deacylase enzymes (SIRT1 through SIRT7) that remove acetyl and other acyl groups from lysine residues on histones and non-histone substrates. Each catalytic turnover consumes one NAD+ molecule, cleaving the glycosidic bond and releasing nicotinamide plus O-acetyl-ADP-ribose. Published studies suggest that SIRT1 deacetylates PGC-1α to stimulate mitochondrial biogenesis, SIRT3 targets matrix enzymes including SOD2 and acetyl-CoA synthetase 2, and SIRT6 regulates telomeric chromatin and glucose homeostasis. Because sirtuin activity is bounded by NAD+ concentration, any decline in cellular NAD+ translates directly into reduced deacetylation capacity. Imai and Guarente summarised this dependency in their 2014 Trends in Cell Biology review (PMID 24786309), framing NAD+ as a rate-limiting substrate for the entire sirtuin family in mammalian cell-culture and rodent models.
How does PARP activity compete with sirtuins for NAD+?
Poly(ADP-ribose) polymerases, especially PARP1, detect DNA strand breaks and catalyse the transfer of ADP-ribose units onto target proteins — a modification that recruits repair machinery. Each ADP-ribosylation event consumes NAD+, and a single PARP1 activation episode can deplete a large fraction of nuclear NAD+ within minutes. Preclinical research indicates that accumulating DNA damage with age chronically elevates PARP1 activity, siphoning NAD+ away from sirtuins and other consumers such as CD38 and SARM1. A 2017 Science report by Li et al. described how DBC1 binding to PARP1 links NAD+ availability to repair efficiency in murine tissue, an observation frequently cited in aging-focused laboratory protocols that use the 500mg NAD+ research format for in-vitro supplementation studies.
What does published research suggest about NAD+ decline with age?
Multiple quantitative studies have measured tissue NAD+ across the lifespan. Massudi and colleagues (PLOS ONE, 2012) reported that human skin NAD+ fell by roughly half between young adulthood and old age. A 2018 Nature Communications paper by Zhou et al. documented parallel declines in hepatic NAD+ alongside rising CD38 expression in aged rodents, implicating the NADase activity of CD38 as a major contributor. Investigators studying the hallmarks of aging framework treat NAD+ decline as an upstream driver of mitochondrial dysfunction, chronic inflammation, and impaired nutrient sensing. Published reviews suggest that restoring NAD+ pools via precursor supplementation — nicotinamide riboside, nicotinamide mononucleotide, or direct NAD+ — recovers sirtuin-dependent transcriptional programmes in preclinical models, though responses vary by tissue and animal age.
Why is the 500mg research format useful for laboratory protocols?
NAD+ studies often require larger masses than typical peptide experiments because molar potency at cellular targets (sirtuins, CD38) falls in the micromolar-to-millimolar range rather than the nanomolar range typical of receptor-binding peptides. A 500 mg vial delivers roughly 0.75 millimoles of pure coenzyme, sufficient for multi-plate cell-culture titrations, enzymatic assays of isolated sirtuins, or multi-day perfusion experiments on isolated tissue. The NAD+ 500mg research format ships as lyophilised powder, which protects the labile glycosidic bond during transit and storage. Researchers running comparison experiments between NAD+ and mitochondrial peptides frequently standardise on this vial size so that a single aliquoting workflow covers both the coenzyme arm and any peptide arm of a protocol. A broader discussion of mitochondrial-targeted comparisons is available in our NAD+ vs SS-31 research comparison.
How is NAD+ reconstituted for laboratory use?
NAD+ dissolves readily in bacteriostatic water, sterile water for injection, or phosphate-buffered saline at neutral pH. A common laboratory working stock is 100 mM, prepared by dissolving 66.3 mg of NAD+ in 1 mL of vehicle. Because the pyridinium ring is hydrolytically labile, reconstituted solutions are best prepared fresh, filter-sterilised through a 0.22 µm membrane, and used within 24 hours, or flash-frozen in single-use aliquots at -80°C. Preclinical research indicates that freeze-thaw cycles progressively degrade the glycosidic bond; investigators measuring intact NAD+ by HPLC or fluorimetric assay should verify each lot after long storage. Lyophilised powder at -20°C, protected from moisture in a desiccator, retains activity for years under documented stability studies. All handling steps are performed for laboratory and research purposes only.
What cell-culture models dominate NAD+ research?
HepG2 hepatocytes, C2C12 myoblasts, HEK293 cells, and primary murine embryonic fibroblasts appear repeatedly in the NAD+ literature because each expresses a tractable complement of sirtuins and NAD+ biosynthetic enzymes. Primary cardiomyocytes and isolated mitochondria are the preferred systems for investigators focused on oxidative phosphorylation. Published studies suggest that the NAD+/NADH ratio measured by mass spectrometry tracks closely with metabolic phenotype in these models, so researchers often pair NAD+ treatment arms with isotopologue tracing or Seahorse respirometry. In aged-mouse models of sarcopenia, neurodegeneration, and metabolic decline, investigators measure tissue NAD+ by LC-MS/MS and correlate outcomes with sirtuin target acetylation status — for example, mitochondrial SOD2 K68 acetylation as a SIRT3 readout.
How do CD38 and SARM1 shape NAD+ turnover?
CD38 is a transmembrane NADase whose expression rises with age and inflammation, hydrolysing NAD+ to generate cyclic ADP-ribose and nicotinamide. A 2016 Cell Metabolism paper by Camacho-Pereira et al. demonstrated that CD38-knockout mice maintain youthful NAD+ levels and mitochondrial function in aged liver and muscle. SARM1, a toll-interleukin-1-receptor-domain protein, activates catastrophic NAD+ cleavage during axonal injury and has become a focus of neurodegeneration research. Preclinical research indicates that CD38 inhibitors and SARM1 antagonists preserve NAD+ pools in parallel with precursor supplementation, and laboratory protocols often combine enzyme-inhibition arms with direct NAD+ supplementation to dissect contributions of synthesis versus degradation pathways in the aged phenotype.
Ready to source NAD+ for your research protocol?
Investigators purchasing for laboratory use can acquire the NAD+ 500mg research vial directly from Peptideware. Each lot is lyophilised, third-party verified for identity and purity, and shipped with a certificate of analysis. For comparative work alongside mitochondrial-targeted peptides, see our NAD+ vs SS-31 research comparison and the catalogue-wide longevity peptides guide.
Frequently Asked Questions
Is NAD+ a peptide?
No. NAD+ is a dinucleotide coenzyme composed of nicotinamide mononucleotide linked to adenosine monophosphate through a pyrophosphate bridge — a nucleotide-based structure rather than an amide-linked chain of amino acids. Peptideware catalogues it alongside peptide compounds because investigators studying mitochondrial and longevity peptides routinely examine NAD+ pools in parallel experimental arms. The molecule participates in hundreds of redox reactions and serves as an obligate substrate for sirtuin deacylases and PARP polymerases, which is why it appears so often in preclinical research on aging, metabolism, and DNA repair. When labelling samples or preparing protocol documents, classify NAD+ as a coenzyme rather than a peptide to avoid downstream confusion with peptide-specific handling practices and reconstitution norms.
What is the molecular weight of NAD+?
Oxidised NAD+ has a molecular weight of 663.43 g/mol with the chemical formula C21H27N7O14P2. The reduced form NADH weighs 665.44 g/mol because of the additional hydride. Researchers preparing millimolar working stocks typically dissolve 66.3 mg of NAD+ powder in 1 mL of bacteriostatic water or PBS at neutral pH to yield a 100 mM solution. A 500 mg vial therefore contains approximately 0.754 millimoles — sufficient for multiple large-scale cell-culture titrations or enzymatic assays. Because the glycosidic bond is hydrolytically labile, solutions should be prepared immediately before use or flash-frozen in single-use aliquots at -80°C. Researchers confirming concentration typically rely on UV absorbance at 260 nm with the established molar extinction coefficient for the oxidised form.
How does NAD+ differ from NMN and NR precursors?
Nicotinamide mononucleotide (NMN) and nicotinamide riboside (NR) are biosynthetic precursors that cells convert into NAD+ through the salvage pathway. NMN is a single nucleotide that enters cells via the Slc12a8 transporter (or after extracellular conversion to NR) and is phosphorylated and adenylated to yield NAD+. NR enters cells via nucleoside transporters and is phosphorylated by NRK1/2 to become NMN. Preclinical research indicates that each precursor raises intracellular NAD+ pools in mammalian models, though efficacy varies by tissue and dosing vehicle. Direct NAD+ supplementation bypasses these biosynthetic steps entirely, which is useful in cell-free enzymatic assays and in membrane-permeable preparations for isolated mitochondria, where the investigator requires immediate substrate availability without biosynthetic lag.
Why does laboratory research focus on NAD+ and aging?
Published studies suggest that tissue NAD+ pools decline measurably with age across mammalian models — skin, liver, skeletal muscle, brain, and vasculature all show reduced concentrations in aged versus young animals. Because NAD+ is the obligate substrate for sirtuin deacylases, PARP polymerases, and cADPR synthases, its decline ripples across mitochondrial biogenesis, DNA repair, and calcium signalling simultaneously. Aging research frameworks such as the hallmarks of aging position NAD+ depletion as an upstream driver that connects multiple downstream dysfunctions. Laboratory protocols examining whether NAD+ restoration reverses mitochondrial deficits, epigenetic drift, or stem-cell exhaustion have been published across Cell, Science, Nature, and Cell Metabolism, establishing the coenzyme as a central node in preclinical longevity research.
What storage conditions preserve NAD+ activity?
Lyophilised NAD+ powder is stable for years at -20°C when protected from moisture in a sealed, desiccated container. Short-term storage at 2-8°C is acceptable for unopened vials in transit. Once reconstituted in aqueous buffer, the coenzyme should be used within 24 hours at 2-8°C or flash-frozen in single-use aliquots at -80°C to limit hydrolysis of the glycosidic bond. Repeated freeze-thaw cycles progressively degrade activity, so researchers typically aliquot immediately after reconstitution. Solutions held near physiological pH (7.0-7.4) are more stable than those stored in strongly acidic or alkaline conditions. Investigators running longitudinal experiments should verify stock concentration periodically by UV absorbance at 260 nm before each experimental arm.
Which sirtuins are most responsive to NAD+ supplementation in research models?
SIRT1 and SIRT3 are the two sirtuins most frequently documented as responsive to NAD+ availability in preclinical research. SIRT1 resides primarily in the nucleus and deacetylates PGC-1α, FOXO transcription factors, and p53, linking NAD+ status to mitochondrial biogenesis and stress resistance programmes. SIRT3 sits in the mitochondrial matrix and targets SOD2, acetyl-CoA synthetase 2, and multiple oxidative-phosphorylation components. SIRT6 participates in chromatin regulation and base-excision repair and has emerged as a focus of aging research, though its in-vivo NAD+ sensitivity is more complex. Researchers selecting readout endpoints typically pair SIRT1 activity with PGC-1α acetylation status and SIRT3 activity with SOD2 K68 acetylation by Western blot in their cell-culture and rodent-tissue protocols.
How is NAD+ quantified in research samples?
The gold standard for NAD+ quantification is liquid chromatography with tandem mass spectrometry (LC-MS/MS), which separates NAD+ from NADH, NMN, and NADP+ and reports absolute concentrations from isotope-labelled internal standards. Enzymatic cycling assays using alcohol dehydrogenase or diaphorase provide a cheaper fluorometric readout sufficient for relative comparisons. Commercial kits using the WST-1 tetrazolium readout deliver a 96-well plate format for screening, though these kits typically measure total NAD+ plus NADH unless an alkaline preparation step destroys the reduced form. Investigators comparing laboratory protocols across publications should note which method was used, because absolute concentrations vary substantially between LC-MS/MS and cycling-assay reports even for the same tissue type from the same animal strain.
Research Disclaimer: All products are intended for laboratory and research purposes only. Not for human or animal consumption. These statements have not been evaluated by the FDA. The content above summarises preclinical and in-vitro research literature and does not constitute medical advice or recommendations.