Last Updated: April 14, 2026
Among growth hormone (GH) secretagogues studied in preclinical research, ipamorelin vs CJC-1295 represents one of the most frequently examined pairings in the literature. Both compounds act on the hypothalamic-pituitary axis to stimulate endogenous GH release from pituitary somatotrophs, yet they do so through distinct receptor systems with markedly different pharmacokinetic profiles. Ipamorelin is a synthetic pentapeptide classified as a growth hormone-releasing peptide (GHRP) and selective agonist of the ghrelin receptor (GHSR-1a). CJC-1295, by contrast, is an analog of growth hormone-releasing hormone (GHRH) that binds the GHRH receptor on somatotroph cells. Published studies suggest that combining these two mechanistic classes produces a synergistic amplification of GH pulses in animal models, making the pair a common reference protocol in longevity and somatotroph physiology research. This comparison examines receptor targets, half-life differences, selectivity, and the body of preclinical research supporting each compound for laboratory use.
Quick Comparison Table
| Attribute | Ipamorelin | CJC-1295 (No DAC) | CJC-1295 (DAC) |
|---|---|---|---|
| Class | GHRP / ghrelin receptor agonist | GHRH analog | GHRH analog with albumin binder |
| Receptor | GHSR-1a | GHRH-R | GHRH-R |
| Structure | Pentapeptide (5 aa) | Modified GHRH(1-29) | GHRH(1-29) + DAC (Drug Affinity Complex) |
| Half-life (preclinical) | ~2 hours | ~30 minutes | ~8 days |
| GH release pattern | Pulsatile, ghrelin-mimetic | Pulsatile, physiologic | Sustained GH elevation |
| Selectivity | Minimal cortisol / prolactin / ACTH | GHRH-R specific | GHRH-R specific |
| Research pairing | Often combined with GHRH analogs | Often combined with GHRPs | Often combined with GHRPs |
What Is a GH Secretagogue?
A growth hormone secretagogue is any compound that stimulates pituitary somatotrophs to release endogenous GH rather than delivering exogenous GH directly. Preclinical research divides this class into two distinct mechanistic families that act on separate receptor systems and produce complementary downstream signaling.
The first family is the GHRH (growth hormone-releasing hormone) analog group. Natural GHRH is a 44-amino-acid hypothalamic neuropeptide, but the bioactive portion resides in the first 29 residues. GHRH analogs like CJC-1295 and sermorelin bind the GHRH receptor (GHRH-R), a Class B G-protein-coupled receptor, activating cyclic AMP signaling and increasing somatotroph sensitivity to GH-releasing signals.
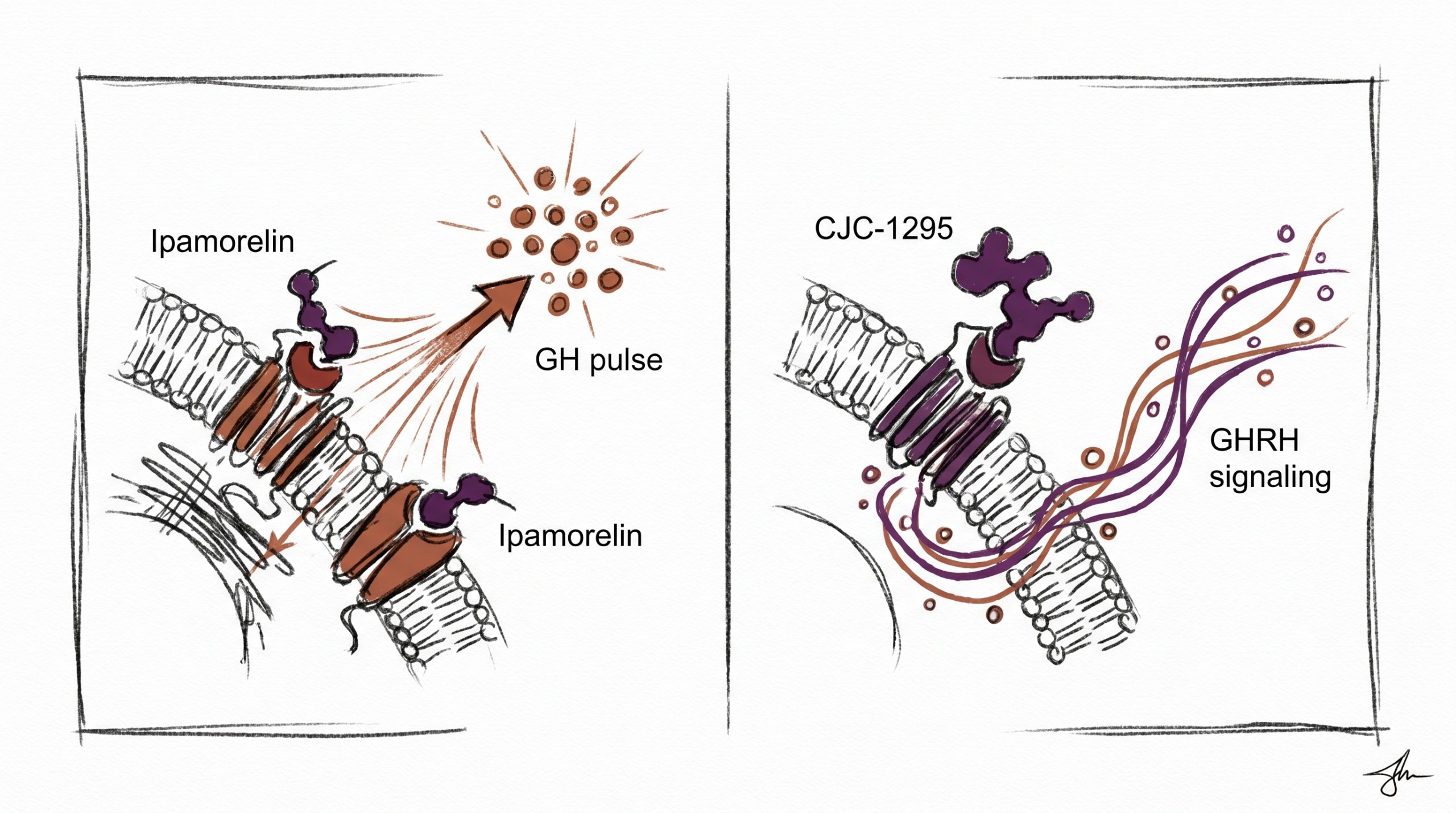
The second family is the GHRP (growth hormone-releasing peptide) group, which includes ipamorelin, GHRP-2, GHRP-6, and hexarelin. These compounds mimic ghrelin by binding the growth hormone secretagogue receptor (GHSR-1a) on somatotrophs and hypothalamic neurons. This pathway functions independently of GHRH-R and amplifies GH pulses through a complementary cAMP-independent mechanism involving phospholipase C signaling. Because the two families act at separate receptors, preclinical research frequently examines them together to study additive or synergistic effects on pulsatile GH release.
How Does Ipamorelin Work?
Ipamorelin is a synthetic pentapeptide (Aib-His-D-2-Nal-D-Phe-Lys-NH2) developed in the late 1990s and characterized as a “third generation” GHRP in published literature. It binds selectively to the ghrelin receptor GHSR-1a on pituitary somatotrophs, triggering calcium mobilization and GH release through phospholipase C activation.
What distinguishes ipamorelin from earlier GHRP analogs like GHRP-2 and GHRP-6 is its selectivity profile. Preclinical research indicates that ipamorelin produces robust GH release with minimal activation of secondary pathways that release cortisol, prolactin, or adrenocorticotropic hormone (ACTH). In the 1998 paper by Raun et al. that first characterized the compound (European Journal of Endocrinology), ipamorelin stimulated GH release in rat and swine models at potencies comparable to GHRP-6 while showing no measurable prolactin or ACTH elevation at GH-stimulating doses. This selectivity has made it a preferred reference compound in studies isolating GHSR-1a effects from broader endocrine activation.
Ipamorelin has a reported plasma half-life of approximately two hours in preclinical models, which is substantially longer than ghrelin itself (~10 minutes) but short enough that pulsatile dosing protocols retain a physiologic release pattern. Research-grade Ipamorelin No DAC 10mg is commonly used in laboratory protocols examining somatotroph responsiveness, hypothalamic-pituitary axis feedback, and combination studies with GHRH analogs.
How Does CJC-1295 Work?
CJC-1295 is a modified analog of GHRH(1-29), the biologically active N-terminal fragment of human GHRH. The parent sequence is rapidly degraded in circulation by dipeptidyl peptidase-IV (DPP-IV), which cleaves the Tyr-Ala bond at position 2. CJC-1295 incorporates four amino acid substitutions (D-Ala at position 2, Gln at 8, Ala at 15, Leu at 27) that confer resistance to DPP-IV cleavage and extend plasma stability.
Two distinct variants of CJC-1295 appear in the research literature, and the distinction is critical for experimental design:
- CJC-1295 No DAC (also called modified GHRH(1-29) or “MOD GRF 1-29”) contains only the four stabilizing substitutions. Its half-life in preclinical models is approximately 30 minutes — long enough to resist DPP-IV degradation but short enough to preserve the natural pulsatile pattern of GH release. Research-grade CJC-1295 No DAC 5mg is commonly used in pulsatile protocol studies.
- CJC-1295 DAC (Drug Affinity Complex) adds a maleimidopropionic acid (MPA) group that covalently binds to serum albumin via cysteine-34, producing a bioconjugate with an extended half-life of approximately 8 days in preclinical models. This sustained exposure produces a “GH bleed” — a continuous elevation of circulating GH and IGF-1 rather than pulsatile release. Research-grade CJC-1295 DAC 10mg is used in studies where sustained receptor occupancy is the experimental variable of interest.
The choice between DAC and No DAC depends entirely on whether the research protocol is modeling pulsatile versus tonic GH exposure. Published preclinical studies suggest these two exposure patterns produce materially different downstream effects on IGF-1 kinetics, receptor desensitization, and hepatic gene expression.
What Is the “Amplifier Effect”?
The term “amplifier effect” in GH secretagogue research describes the synergistic GH release observed when a GHRP and a GHRH analog are co-administered. The phenomenon was characterized in preclinical and human subject research beginning in the 1990s and is mechanistically grounded in the non-overlapping receptor pathways of the two compound classes.
A GHRH analog like CJC-1295 No DAC binds GHRH-R and increases cAMP-mediated GH synthesis and release readiness. A GHRP like ipamorelin binds GHSR-1a and triggers calcium-mediated GH secretion while also suppressing somatostatin tone (somatostatin is the primary inhibitory brake on GH release). When both receptors are activated simultaneously, research indicates that the resulting GH pulse is substantially larger than the sum of either compound administered alone. Published studies summarizing these findings include work by Bowers and colleagues on the discovery of GHRPs (Journal of Clinical Endocrinology & Metabolism, 1990s).
In research contexts, this synergy is why ipamorelin and CJC-1295 No DAC are frequently reconstituted and examined together. The pairing allows investigators to study maximal somatotroph responsiveness while preserving a pulsatile release pattern that more closely mirrors endogenous GH rhythms in preclinical models.
What Does Published Research Show?
The peer-reviewed literature on these two compounds spans discovery pharmacology, receptor selectivity characterization, and applied somatotroph physiology studies.
Ipamorelin literature: The foundational characterization by Raun et al. (1998, European Journal of Endocrinology) established ipamorelin as a potent and selective GH secretagogue with no measurable effect on prolactin, ACTH, or cortisol at GH-effective doses in preclinical models. Subsequent research has examined GHSR-1a binding affinity, downstream signaling via Gq/phospholipase C, and comparative potency against ghrelin and other GHRPs. Studies indexed in PubMed include investigations into bone turnover markers, gastrointestinal motility (a separate GHSR-1a-mediated effect), and combination pharmacology with GHRH analogs in rodent models.
CJC-1295 literature: Teichman et al. (2006, Journal of Clinical Endocrinology & Metabolism) reported the pharmacokinetic and pharmacodynamic characterization of CJC-1295 DAC, documenting the sustained elevation of GH and IGF-1 over multiple days following single administration. Earlier work on the No DAC variant (often referred to as “modified GRF(1-29)”) established DPP-IV resistance and receptor selectivity. Preclinical research indicates that the two variants produce distinct IGF-1 response curves: pulsatile with No DAC, sustained-plateau with DAC.
Combination research: Studies examining GHRP + GHRH co-administration report synergistic GH pulse amplitudes in both rodent and primate preclinical models, with the effect typically exceeding the arithmetic sum of individual compound responses by a factor of two or more in reported ranges.
Which Should Researchers Choose?
Selection between ipamorelin, CJC-1295 No DAC, and CJC-1295 DAC depends on the experimental question under investigation.
For isolated GHSR-1a pathway studies: Ipamorelin alone provides a selective probe of the ghrelin receptor arm of GH release without confounding cortisol or prolactin responses that complicate data interpretation with older GHRPs.
For pulsatile GHRH-R studies: CJC-1295 No DAC offers DPP-IV resistance while preserving the short half-life needed to maintain physiologic GH pulse architecture. This is the appropriate choice when the research model requires endogenous-pattern release.
For sustained GH exposure studies: CJC-1295 DAC is used when the experimental variable is tonic rather than pulsatile receptor occupancy. The ~8-day half-life makes it appropriate for multi-day exposure protocols but unsuitable for any study modeling physiologic pulsatility.
For maximal GH pulse amplitude (amplifier effect) studies: A research protocol combining ipamorelin with CJC-1295 No DAC is the most common reference stack in published literature. Both compounds are reconstituted for research according to standard laboratory procedures and examined in parallel to capture the synergistic pulse.
For broader context on GH secretagogues and related longevity-focused research compounds, see the Longevity Peptides Research Guide.
Frequently Asked Questions
Why are ipamorelin and CJC-1295 combined in research protocols?
The two compounds act on separate receptors — ipamorelin on GHSR-1a (the ghrelin receptor) and CJC-1295 on GHRH-R (the GHRH receptor) — and their combined activation produces a synergistic amplification of growth hormone release that exceeds the sum of either compound administered alone. Preclinical research indicates that simultaneous stimulation of both pathways drives larger GH pulses because the two receptors engage complementary intracellular signaling cascades (calcium-phospholipase C versus cAMP), and ipamorelin additionally suppresses somatostatin inhibitory tone on the somatotroph. This synergy is commonly referred to as the “amplifier effect” in published literature, and it is the primary mechanistic rationale for pairing the two compounds in laboratory protocols examining maximal somatotroph responsiveness.
What does “third generation GHRP” mean when applied to ipamorelin?
In growth hormone secretagogue research, “generation” refers to successive waves of synthetic GHRP analogs with improved selectivity profiles. First-generation GHRPs like GHRP-6 stimulate GH release but also elevate cortisol, prolactin, and appetite through secondary pathways. Second-generation compounds like GHRP-2 and hexarelin improved potency but still produced measurable prolactin and cortisol responses in preclinical models. Ipamorelin is classified as third generation because published characterization studies (notably Raun et al. 1998) demonstrated robust GH release at GHSR-1a with essentially no activation of cortisol, ACTH, or prolactin pathways at GH-effective doses. This selectivity makes ipamorelin a cleaner research tool for isolating GHSR-1a-specific effects from broader endocrine activation in laboratory settings.
How are ipamorelin and CJC-1295 reconstituted together for research?
Each lyophilized peptide is reconstituted separately using bacteriostatic water or sterile water in standard laboratory protocols, with final concentrations typically expressed in mcg per 0.1 mL for reference consistency across studies. After reconstitution, the two compounds can be drawn into the same research solution or maintained in separate vials depending on the protocol. Storage is refrigerated at 2–8°C after reconstitution for short-term research use, with stability reported in published stability data for both compound classes. All handling is conducted under standard laboratory practice with appropriate personal protective equipment. These products are intended for laboratory and research purposes only and are not for human or animal consumption. Consult published reconstitution protocols before beginning any experimental work.
What is the difference between CJC-1295 with DAC and without DAC?
The difference is the addition of a Drug Affinity Complex (DAC) — a maleimidopropionic acid moiety that covalently binds serum albumin via cysteine-34 once the peptide enters circulation. Without DAC, CJC-1295 is simply a DPP-IV-resistant GHRH(1-29) analog with a preclinical half-life of approximately 30 minutes, producing pulsatile GH release consistent with physiologic rhythms. With DAC, the albumin conjugate extends the circulating half-life to approximately 8 days in preclinical models, producing sustained elevation of GH and IGF-1 over multiple days rather than discrete pulses. The two variants are appropriate for fundamentally different research questions — pulsatile studies use No DAC, while sustained exposure studies use DAC. They are not interchangeable in experimental design.
Does ipamorelin affect cortisol or prolactin like older GHRPs?
Preclinical research indicates that ipamorelin produces minimal to undetectable cortisol, prolactin, and ACTH responses at doses that robustly stimulate GH release. This selectivity was a defining finding in the original Raun et al. characterization published in European Journal of Endocrinology in 1998 and has been confirmed in subsequent receptor binding and endocrine response studies. By contrast, earlier GHRPs like GHRP-6 produce measurable cortisol and prolactin elevations alongside GH release, and GHRP-2 retains some prolactin activity. For researchers designing experiments where the GH response must be isolated from broader hypothalamic-pituitary-adrenal axis activation, ipamorelin’s selectivity profile is the primary reason it is frequently preferred as a GHSR-1a research probe over older GHRP analogs.
Why does the CJC-1295 DAC produce a “GH bleed” instead of pulses?
Endogenous GH release is pulsatile — discrete bursts triggered by hypothalamic GHRH signals, with intervening periods of low circulating GH due to somatostatin inhibition. When a GHRH analog is circulating continuously at pharmacologic concentrations for many days (as occurs with CJC-1295 DAC’s ~8-day half-life), the somatotroph is under persistent stimulation and GH release becomes tonic rather than pulsatile — the so-called “GH bleed.” Published preclinical research suggests that this sustained exposure pattern produces materially different downstream effects compared with pulsatile release, including different IGF-1 kinetics and altered hepatic gene expression patterns. This is why DAC and No DAC variants are treated as distinct research tools answering different experimental questions, not as interchangeable forms of the same compound.
All products are intended for laboratory and research purposes only. Not for human or animal consumption. These statements have not been evaluated by the FDA.